Geochemistry: Definition, History, Methods, and Earth’s Composition
Geochemistry is the study of the distribution and abundance of chemical elements in minerals, rocks, ore bodies, rock units, soils, the Earth, atmosphere, and by some accounts, other celestial bodies, along with the principles governing the distribution and migration of these elements. This branch of Earth science encompasses the analysis of chemical element movement, the properties of minerals related to their elemental concentrations, and the classification of rocks based on chemical composition. The field began with the discovery of 31 chemical elements by French chemist Antoine Lavoisier in 1789, and the term “geochemistry” was first mentioned by German chemist Christian F. Schönbein in 1813.
In 1884, the United States Geological Survey (USGS) established a laboratory to investigate planetary chemistry, appointing F. W. Clarke as its head; since then, the USGS has remained a global leader in geochemical data collection and analysis. In 1904, the Carnegie Institution in Washington, D.C., founded the Geophysical Laboratory to test physical and chemical properties of minerals and rocks, while the Vernadsky Institute in Moscow, Russia, had a similar mission; both institutions spearheaded technological revolutions in rock composition analysis, leading to concepts of chemical equilibrium and disequilibrium and the amassing of vast global rock chemistry databases. Geochemist Victor M. Goldschmidt from the University of Oslo, Norway, applied the phase rule to explain metamorphic changes in terms of chemical equilibrium.
Geochemistry is commonly studied using several complementary methods. Chemical analysis involves breaking down rocks into major and minor (trace) constituents and measuring them in the laboratory. Analysis of atomic and chemical structure examines the physical properties of minerals and rocks as reflected in their geochemistry. Direct experimentation under controlled conditions simulates formation processes of various materials and determines phase relations between them. Additionally, the study of dispersion and accumulation of elements under dynamic conditions provides insights into element behavior in natural systems.
The discipline is subdivided into several specialized fields. Isotope geochemistry measures the concentration of elements and their isotopes in different Earth and planetary systems, using stable and radiogenic isotopes to understand mineral and rock systems, and dates geological events via radioactive decay. Geochemical cycling tracks changes in element distribution across Earth’s compartments to understand mineral, rock, water, and biological systems. Cosmochemistry analyzes the distribution of elements and their isotopes in space. Biogeochemistry assesses the role of life and organisms on Earth’s chemistry and Earth systems. Organic geochemistry examines processes and compounds derived from living and once-living organisms. Environmental and exploration geochemistry applies chemical principles to environmental, hydrological, and mineral exploration studies.
Geochemistry and the Composition of the Earth. Geochemists have collaborated closely with cosmologists to better understand the chemical composition of the universe and the Earth-Moon system within cosmochemistry. These cosmochemists have developed models showing that the universe’s chemical composition began almost exclusively with hydrogen and helium and evolved through stellar processes and supernovae to include heavier elements. Studies of meteorites have yielded data on the average composition of the inner solar system’s rocky planets, and these data have been used to derive a model for Earth’s average composition. Earth is thought to have an overall composition close to that of a carbonaceous chondrite meteorite. The planet experienced early heating due to the decay of short-lived radioactive isotopes, heat from gravitational compaction, and the collection of metallic phases in the core that sank and released heat. This early heating formed a melt phase within Earth, and the outer core remains molten today, as demonstrated by seismic waves, resulting in chemical and density zonation within the planet.
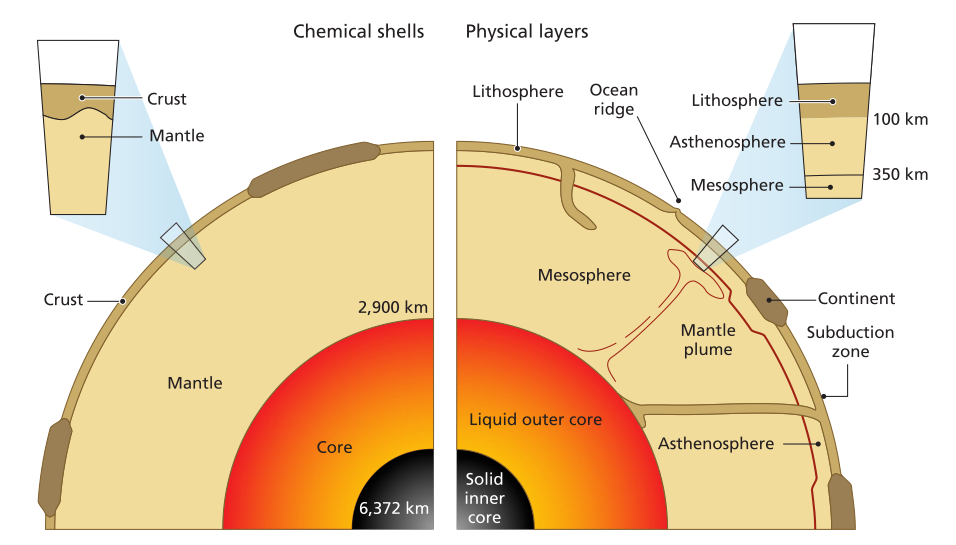
Cross sections of the Earth showing chemical shells (crust, mantle, and core) and physical layers (lithosphere, asthenosphere, mesosphere, outer core, and inner core)
The chemical and density zonation has divided Earth into several distinct shells with different properties: the crust, upper mantle, transition zone, lower mantle, outer core, and inner core. Earth’s magnetic field is generated by motions in the liquid outer core, where a dynamo effect arises from the movement of electrically charged fluid. The lower mantle has a composition that includes magnesium and iron silicate concentrations, similar to chondritic meteorites. Several major polymorphic transitions occur within Earth that are crucial for determining its overall structure and behavior. At 250 miles (400 km) depth, pyroxene minerals adopt a garnet structure due to increased pressure while maintaining a pyroxene composition. Simultaneously, the crystal lattice structure of olivine—one of the most abundant mantle minerals—changes from an olivine to a denser spinel structure. Deeper, at 435 miles (700 km), garnet and spinel structures react together and transform into progressively denser crystal lattices, including ilmenite and then a perovskite structure, still retaining olivine and pyroxene compositions though possibly with more iron present.
Geochemists have determined that Earth is not yet completely chemically fractionated. Volcanoes continue to release gases such as sulfur and carbon dioxide, and deep eruptions, such as those through kimberlites, have recently released diamonds, indicating that carbon still exists at depth. Carbon dioxide is presently being degassed from Earth’s interior at high rates, showing that fractionation is an ongoing process. The composition of Earth and its different layers has been calculated based on density measurements, gravity, deep samples erupted from volcanoes, and models based on meteorite composition. Geochemists have calculated the crust’s composition as approximately: 47% oxygen (O), 28% silicon (Si), 11% iron (Fe), magnesium (Mg), and calcium (Ca), 8% aluminum (Al), and 6% potassium (K), sodium (Na), and all other elements. In terms of minerals in Earth’s crust, this equates to about 49% feldspar, 21% quartz, 5% pyroboles (including pyroxene, amphibole, and olivine), 8% micas, and 7% magnetite and all other minerals.
The overall distribution of elements in the whole Earth is quite different and resembles a massively differentiated giant meteorite with a crust on top. The whole Earth has a composition comprising: 38% of its mass in the core, divided between 35% iron (Fe) and 2.7% nickel (Ni); 28% oxygen, distributed mostly in the mantle; 17% magnesium (Mg); 13% silicon (Si); and 2.7% sulfur (S).
Further Reading: Holland, H. D., and K. K. Turekian. Treatise on Geochemistry. 9 Vols. Amsterdam: Elsevier, 2004.
Marshall, C., and R. Fairbridge. Encyclopedia of Geochemistry. Berlin: Springer, 2006.
Date added: 2026-04-26; views: 2;
