Role of Neutrophils and Platelets. Role of MicroRNAs
Neutrophils have evolved a broad-based capacity for biochemically combating infectious organisms by secreting proteases, ROS, and antimicrobial proteins. However, recent investigation also supports multiple roles for neutrophils in atherogenesis. Within the subendothelial space, neutrophils can produce an array of collagenases, elastases, and other matrix metalloproteinases that can hydrolyze and degrade the intercellular matrix material of plaque and its fibrous cap, thereby weakening them and rendering them more prone to rupture.
Neutrophils also elaborate myeloperoxidase and ROS in the subendothelial space which are cytotoxic and oxidize trapped lipoproteins. Neutrophils entering the subendothelial space also potentiate injury by releasing (1) four different subsets of granules containing preformed proteases, pro-oxidative enzymes, and cytokines whose release is precisely timed in response to conditions in the prevailing histologic milieu and (2) and leukotrienes such as LTB4, a potent chemoattractant.
A more recently elucidated pathway by which neutrophils can promote atherogenesis is by forming neutrophil extracellular traps (NETs) (Fig. 2.8). NETs are produced by suicidal neutrophils and represent an extruded reticular structure composed of decondensed chromatin as well as nuclear, granular, and cytosolic proteins. NETs represent a type of mechanism by which endothelial cells can be exposed to sudden, very high concentrations of proinflammatory mediators. NETosis or the process of NET formation can be induced by ROS, cytokines, cholesterol crystals, and activated platelets. In addition to nucleic acids, the molecular constitution of NETs contains a complex proteome which includes histones, proteases, lysosomal cathepsins, α-defensins, and myeloperoxidase, among other proteins and enzymes. NETS are prothrombotic and cytotoxic.
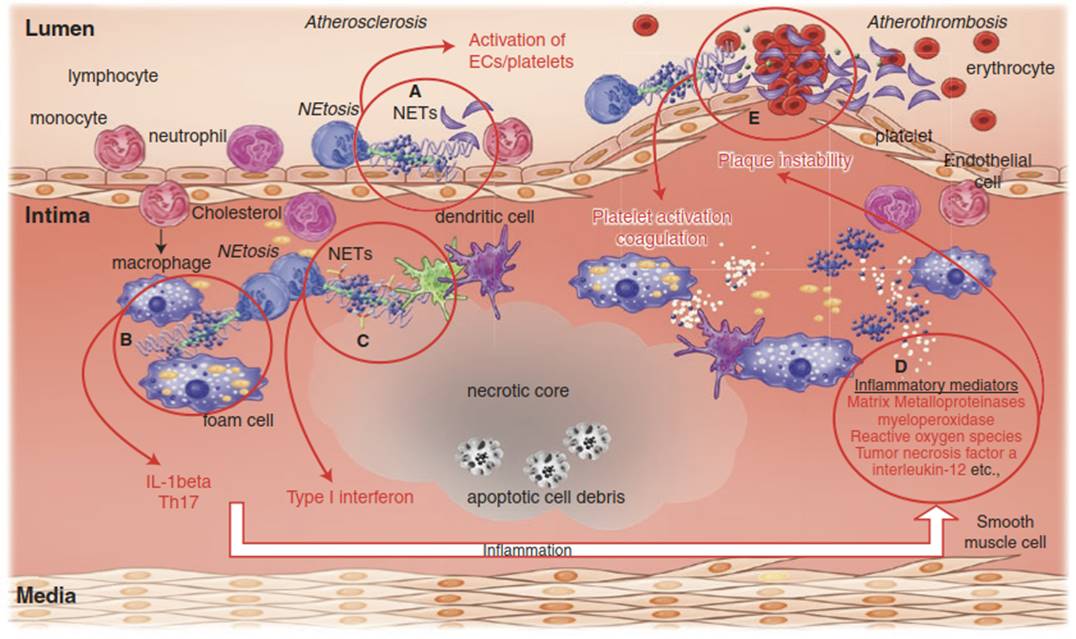
Fig. 2.8. Neutrophil extracellular traps (nets) in atherosclerosis and atherothrombosis. (a) Neutrophils netting in the arterial lumen along the endothelial surface activates endothelial cells, platelets, and other leukocytes, inducing an inflammatory nidus and endothelial dysfunction. (b, c) NETs may stimulate T helper cells to secrete IL-1β and potentiate a type I interferon response, which boosts leukocyte activation and the intensity of inflammation. (d, e) The proinflammatory milieu promotes plaque instability and rupture. In the setting of acute plaque rupture, NETs can participate in thrombus formation by activating the coagulation cascade with overlying thrombus formation and arterial occlusion
Role of Platelets. Platelets are nonnucleated cells arising from parent megakaryocytes and mediate clot formation in concert with coagulation pathways. Platelets potentiate atherogenesis in multiple ways.
Platelet α-granules contain a host of cytokines, chemokines, growth factors, and enzymes that can be mobilized and secreted in response to extracellular stimuli. Platelets interact with endothelial cells and leukocytes according to the following mechanisms:
1. Platelets adhere to dysfunctional endothelial cells by binding to either (a) ICAM-1 via the glycoprotein 2b/3a receptor and fibrinogen or (b) selectin P via glycoprotein 1b.
2. Thrombus formation along the endothelial surface is modulated in a bidirectional manner. Endothelial cells secrete nitric oxide and prostacyclin, which inhibits platelet activation and aggregation. Endothelial cells can also attenuate ADP availability by releasing CD39 ectonucleotidase, which hydrolyzes ADP to AMP and phosphate. Platelets can secrete nitric oxide which inhibits endothelial P-selectin expression, reduces platelet recruitment for clot propagation, and promotes platelet dissociation.
3. Despite being nonnucleate, platelets effectively store messenger RNAs (mRNAs) for subsequent protein translation. In the setting of heightened inflammation, platelets can boost the inflammatory response by releasing IL-1β, among other inflammatory mediators.
3. Inflammation can induce the coactivation of platelets and neutrophils, which leads to increased production of human neutrophil peptide-1 (HNP-1) and regulated activation of normal T cell expressed and secreted (RANTES). RANTES and HNP-1 facilitate monocyte adhesion to endothelial cells and recruitment into the arterial wall.
In addition to signal transmission by cell surface receptors and granule release, platelets can interact with endothelial cells and leukocytes by direct bilateral mRNA transmission, thereby boosting local molecular biosynthetic capacity and an inflammatory response.
6. Platelet microparticles also upregulate the inflammatory response. These microparticles secrete microRNAs (miRNA), which are noncoding RNAs that regulate posttranscriptional gene expression. For example, platelet- derived miRNA-320b decreases surface expression of endothelial ICAM-1 and miRNA-223 stimulates increased phagocytic activity by macrophages resident in the subendothelial space.
Clearly, the interactions of platelets with endothelial cells and other histologic components of the arterial wall and atherosclerotic plaque are complex and highly orchestrated. Much remains to be learned about these processes and how they might be therapeutically modulated.
Role of MicroRNAs. As noted above, miRNAs are noncoding RNAs that regulate posttranscriptional gene expression. MicroRNAs are highly conserved and bind to the 3' untranslated region of messenger RNA (mRNA) transcripts, resulting in “RNA silencing.”
They are produced and secreted by a large variety of cells. MicroRNAs secreted into the circulation are resistant to the activity of plasma RNases, and they can impact the expression of molecules in target cell types. MicroRNAs can be transported in the plasma on microparticles, HDL particles, or bound to the protein Argonaute2. Distinct patterns of circulating miRNAs have been characterized in the setting of myocardial infarction, heart failure, and diabetes mellitus. Specific molecular signatures of miRNAs are also apparent in the setting of CAD.
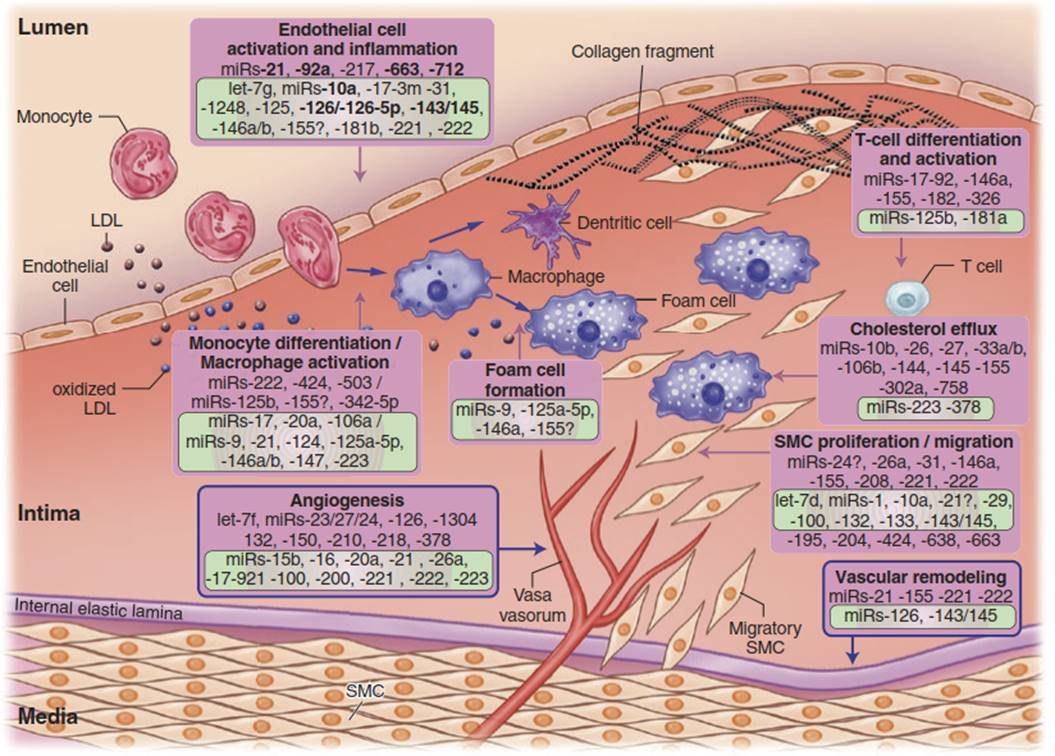
Fig. 2.9 MicroRNAs implicated in atherosclerosis. Positive/atheroprotective (in green frame) or negative/atherogenic (in red frame) effects of miRNAs on atherogen- esis. Question marks indicate controversial or contradictory evidence for specific miRNAs. miRNAs in bold are regulated by blood flow/shear stress. Abbreviations: LDL low-density lipoprotein, SMC smooth muscle cell
The miRNAs do not unexpectedly have a very complex relationship with atherogenesis, with numerous miRNAs that can either stimulate or inhibit expansion of the vasa vasora, macrophage cholesterol efflux, vascular remodeling, smooth muscle cell proliferation and migration, endothelial cell activation, and monocyte and T-cell differentiation and activation, among other functions (Fig. 2.9). Much is yet to be learned about the role of miRNAs in atherogenesis and how the modulation of these regulators of gene expression might be put to therapeutic use.
Date added: 2025-02-17; views: 391;
