Crystal Structure, Defects, and Deformation Mechanisms: A Comprehensive Guide
Crystals represent homogeneous solid structures composed of chemical elements or compounds that possess a regularly repeating arrangement of atoms, alongside a substantial number of crystal lattice defects such as dislocations. All minerals exist as solids, and within each mineral species, atoms organize themselves into a highly regular geometric configuration that remains unique and identical across every specimen of that mineral. This periodic internal arrangement fundamentally determines a mineral's characteristic properties, including its coloration, chemical composition, hardness, and external crystal morphology.
Although many minerals may lack well-developed external crystal faces, they invariably maintain a regularly repeating crystal lattice composed of their constituent atoms. Both external crystal forms and internal atomic arrangements exhibit symmetry, which manifests in several distinct varieties, with crystals displaying four primary symmetry types. Mirror plane symmetry represents the simplest form, wherein an imaginary plane divides the crystal into two halves that are perfect mirror images of one another. Rotational symmetry occurs when a crystal lattice can be rotated about an axis through its center, resulting in identical configurations two, three, four, or six times within a full 360° circuit, corresponding to diads, triads, tetrads, and hexads, respectively.
Roto-inversion constitutes a more complex symmetry operation combining rotation with subsequent inversion of the lattice through its central point. Simple inversion symmetry involves a crystal face positioned diametrically opposite to every other face across the crystal's center. Through various combinations of these symmetry operations, all crystals belong to one of seven crystal systems: cubic, tetragonal, orthorhombic, monoclinic, triclinic, hexagonal, and trigonal.
Despite appearing nearly perfect to the naked eye, crystals invariably contain millions of atomic-scale defects. These imperfections include vacancies within the crystal lattice, various types of atomic arrangement irregularities, and substitutional defects where one atom or ion replaces another of similar charge and size.
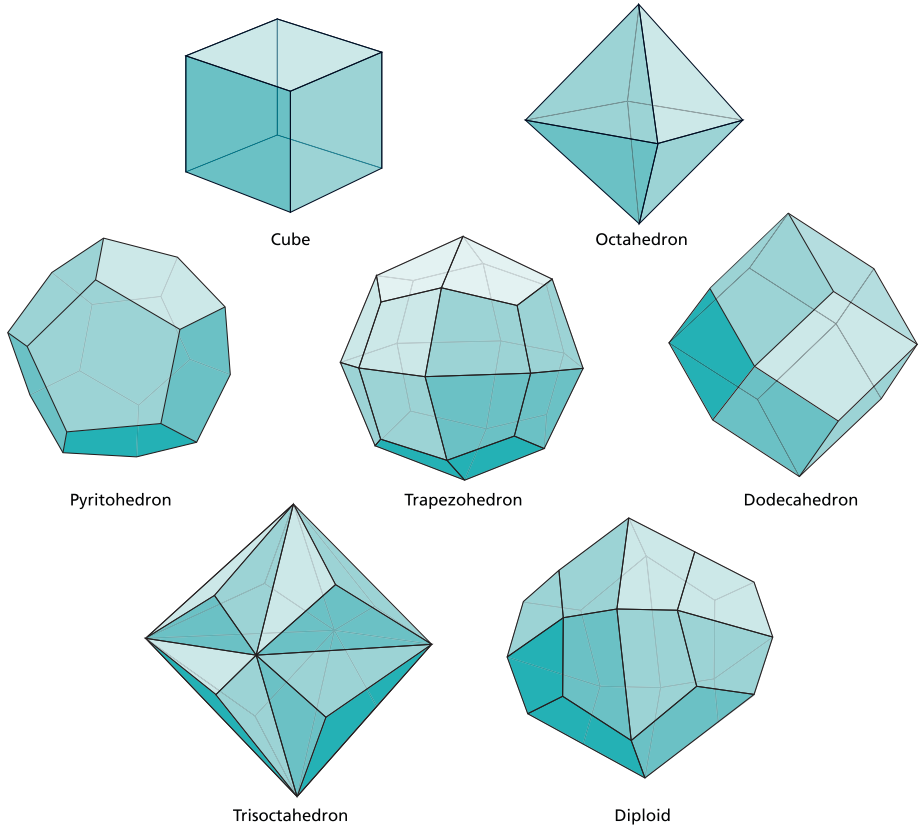
The seven basic types of crystal forms
Crystal Dislocations and Other Defects. Crystals form as regularly ordered, symmetrical arrays of atoms; however, like all natural materials, they remain imperfect and contain numerous defects. Some imperfections arise during initial crystal growth, while others develop subsequently during deformation processes. These defects critically determine the mechanical strength of crystals, minerals, and rocks, and their motion accommodates strain within crystalline materials.
The movement of crystal defects represents a fundamental deformation mechanism, with different defect types exhibiting distinct motion behaviors under varying temperatures, pressures, and applied stresses. Two principal categories of crystal defects exist: point defects and line defects. Point defects encompass vacancies, impurities, and interstitials, whereas line defects are termed dislocations.

Cubic crystal of fluorite, about two inches (5 cm) on each face, on quartz with barite from Frazer’s Hush Mine, Weardale, England (Mark A. Schneider/Photo Researchers, Inc.)
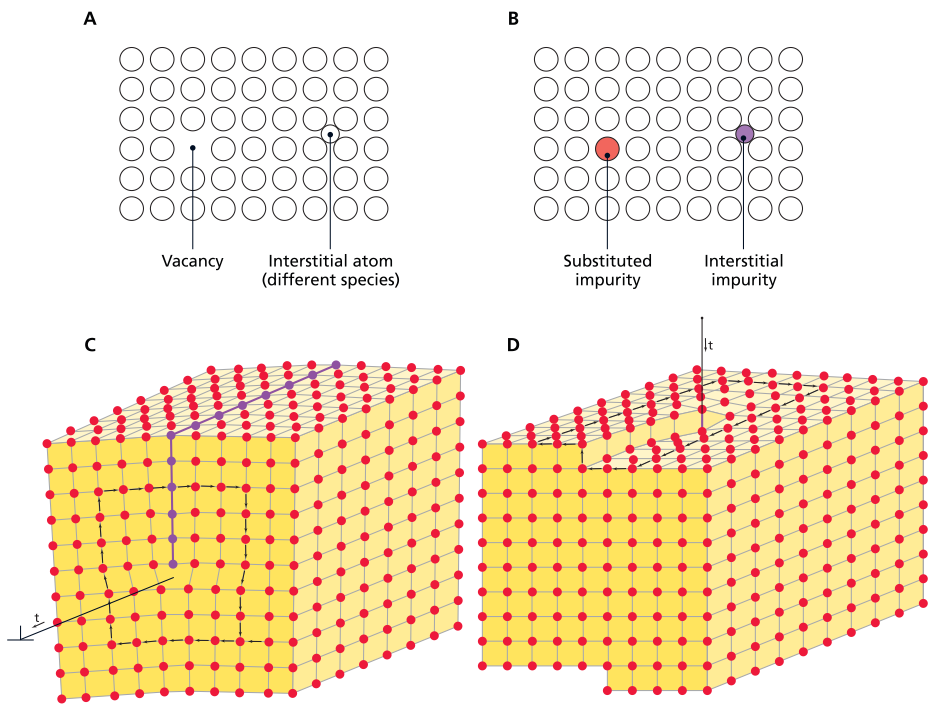
Types of point and line defects in crystals, including vacancies, interstitials, edge dislocation, and screw dislocation
Point Defects in Crystal Lattices. Point defects constitute irregularities affecting a single point within the crystal lattice structure. Impurities occur when an incorrect atom type occupies a lattice site normally reserved for another element. Vacancies represent missing atoms from their expected positions within the atomic lattice. Interstitials involve atoms occupying sites not typically inhabited within the crystal structure. More complex point defect varieties involve multiple atoms and exhibit greater structural complexity.
Within a regularly ordered crystal lattice, most electric charges achieve satisfaction through bonding and the balancing of positive and negative charges. Crystals containing point or other defects possess higher internal energy due to broken or unsatisfied bonds and unbalanced electric charges. Such crystals demonstrate greater reactivity and deformability under stress. Temperature increases promote vacancy formation, whereas elevated pressure reduces vacancy concentrations.
Stress-Induced Diffusion and Twinning. When stress is applied to a crystal, vacancies tend to migrate in an orderly manner relative to the stress field, moving toward crystal faces experiencing the highest stress, while atoms diffuse in the opposite direction toward faces under lowest stress. This vacancy migration process, known as Nabarro-Herring creep, enables general shape changes in crystals, a phenomenon observed during regional deformation in mountain belts worldwide.
Twinning involves the misorientation of a crystallographic plane across a specific lattice plane and can originate from either mismatched growth or applied deformation. Mechanical twinning, also called stress-induced twinning, differs from growth twinning in that shear across a crystal plane alters the lattice orientation. This shear occurs along a crystal plane that must function as a symmetry plane, with the resulting strain limited by crystallographic relationships specific to each crystal type, typically ranging between 20° and 45°.
Translation Gliding and Slip Systems. Translation gliding represents a deformation mechanism wherein the crystal lattice slips along internal crystallographic planes once a critical shear stress threshold is reached. Slip, or translation gliding, preferentially occurs in specific crystallographic directions known as slip systems, favoring directions characterized by short distances between equivalent atoms and orientations that avoid juxtaposing ions of like charge. Slip initiates when the critical resolved shear stress for that crystallographic direction is exceeded.
Most crystals possess multiple slip systems activated at different critical resolved shear stress values, with slip commencing on planes exhibiting the lowest threshold. As deformation progresses, different slip systems begin interfering with one another, causing deformation to either cease or require increased stress for continuation. This stress increase phenomenon is termed work hardening.
To accommodate any general homogeneous strain, crystals require five independent slip systems. Crystals with fewer than five independent slip systems ultimately crack or fracture. Different slip systems may activate under varying temperature conditions or strain rates, making the relationships governing slip system activation under diverse deformation conditions inherently complex.
Dislocation Structure and Behavior. Dislocations are conceptually understood as an extra plane of atoms terminating within the crystal lattice, often referred to as an extra half-plane due to this geometric configuration. The presence of dislocations weakens the crystal structure, and their motion accounts for substantial strain accommodation in crystals. Dislocations move through crystal lattices analogously to how a ridge moves across a carpet, gradually shifting the carpet across a floor without requiring simultaneous movement of the entire structure. The stress needed to break bonds sequentially for dislocation motion is significantly lower than that required for simultaneously breaking all bonds along a specific crystallographic direction.
Two fundamental dislocation types exist: edge dislocations and screw dislocations. Edge dislocations represent the simpler type, essentially constituting an extra half-plane of atoms extending partially across the crystal lattice. Screw dislocations exhibit greater complexity, featuring a twisted geometry analogous to a parking garage structure, where one lattice layer becomes offset by a coiled, screw-like motion about an axis perpendicular to the atomic planes. Most dislocations in natural crystals incorporate both edge and screw components, forming complex geometric configurations. Dislocations create loops within crystals, marking boundaries between slipped and unslipped regions, with this motion and slipping behavior directly analogous to fault movements in geological settings.
Dislocation Interactions. During deformation, numerous dislocations move within crystals, inevitably interacting through several mechanisms. Dislocation annihilation occurs when two dislocations of opposite signs approach each other on the same slip plane, and their extra half-planes meet to form a complete crystal lattice, effectively canceling each other's existence. Each dislocation generates a self-stress field due to disruption of the normal crystallographic structure. These stress fields interact over considerable distances within crystals, causing either repulsion or attraction between dislocations, similar to magnetic interactions. When two dislocations on the same slip plane attract and converge, they annihilate; conversely, repulsion increases dislocation density, requiring progressively higher stresses for continued dislocation motion as deformation proceeds.
Dislocations frequently encounter immobile impurities, particularly interstitials, which effectively pin dislocations behind them. As additional dislocations approach regions containing such impurities, they become similarly trapped and experience repulsion from each other's stress fields, creating dislocation pile-ups. Dislocations may also interact with other dislocations, generating additional pile-up configurations.
Dislocation Motion Mechanisms and Work Hardening. At elevated temperatures during crystal deformation, vacancies can diffuse toward obstacles, or atoms may migrate away from half-planes, enabling dislocations to climb over barriers. Consequently, two primary mechanisms govern dislocation movement through crystals: gliding (planar movement) and climbing (perpendicular movement).
When dislocations from different slip systems intersect, they offset each other's slip planes, forming dislocation jogs—essentially steps within the slip system along which one dislocation traveled. Once a jog exists in a slip plane, dislocation movement along that plane becomes more difficult, requiring climb over the jog for continued progression. Dislocation jogs can be eliminated, or evaporated, through vacancy diffusion toward them or atomic migration away from the jog.
Work hardening encompasses any process rendering further deformation more difficult, necessitating increased stress for equivalent strain. Work hardening occurs through several mechanisms: formation of dislocation jogs, dislocation pile-ups, interaction of stress fields, and increases in dislocation density. This phenomenon predominates at lower temperatures, whereas elevated temperatures promote increased vacancy diffusion and dislocation climb.
Annealing and Recovery Processes. Annealing refers to any process tending to restore a crystal lattice to a less deformed state, primarily through reduction in dislocation numbers. A lattice with fewer dislocations possesses lower energy and greater stability than one with high dislocation density. Common annealing mechanisms include grouping dislocations into more stable configurations, migration of dislocations to crystal edges, and recrystallization of grains. Diffusion facilitates all these processes, making annealing more rapid at elevated temperatures.
Additional annealing mechanisms include opposite-sign dislocations climbing to the same slip plane and annihilating, dislocations gliding and climbing to grain boundaries, formation of subgrain boundaries where dislocation motion concentrates dislocations into planar walls bounding low-dislocation-density domains, and recrystallization involving complete regrowth of the crystal lattice. New grains formed during recrystallization exhibit low dislocation density, with this process typically initiating in regions of high dislocation density or along grain boundaries. Prolonged heating promotes grain growth, where some grains expand at the expense of neighbors, ultimately forming equant grains with 120° grain boundaries at triple junctions.
Alternative Deformation Mechanisms. At temperatures below those facilitating dislocation glide and climb, rocks flow through alternative mechanisms. Pressure solution, also termed grain boundary diffusion or Coble creep, involves crystal flattening and dissolution along edges, with dissolved material either precipitating at grain ends or migrating to form veins, pores, or distant deposits. This deformation mechanism effectively accommodates substantial bulk shortening and extension. Pressure solution frequently produces stylolites—seams representing concentrated insoluble residues from dissolved rock material. In three dimensions, stylolites form irregular surfaces featuring cones or teeth oriented toward the maximum compressive stress direction.
Compaction constitutes another important deformation mechanism, particularly in sedimentary basins where overlying rock weight drives the process. Compaction typically involves dewatering, or fluid expulsion, from rock pore spaces. The weight of newly deposited sediments and overlying rocks increases pressure, forcing grain-to-grain contacts between crystals closer together and expelling interstitial fluids. Some muds initiate with 80 percent porosity and conclude with only 10 percent after burial, expelling substantial water volumes during compaction. Sandstones exhibit initial porosities up to 45 percent, reduced to approximately 10–30 percent depending on rock type, pressure, and fluid conditions, with porosity generally decreasing as burial depth increases.
FURTHER READING: Hull, D., and D. J. Bacon. Introduction to Crystal Dislocations. 3rd ed. Oxford: Pergamon Press, 1984.
Kosevitch, Arnold M. The Crystal Lattice: Phonons, Solitons, Dislocations, Superlattices. 2nd ed. New York: John Wiley & Sons, 2005.
Nabarro, F. R. N. Theory of Crystal Dislocations. New York: Dover, 1987.
Shelly, David. Manual of Optical Mineralogy. Amsterdam: Elsevier, 1980.
Date added: 2026-04-26; views: 2;
