Groundwater Contamination: Arsenic, Sewage, and Seawater Intrusion in Coastal Aquifers
Natural groundwater is typically rich in dissolved elements and compounds derived from the soil, regolith, and bedrock through which the water has migrated. Some of these dissolved elements and compounds are poisonous, whereas others are tolerable in small concentrations but harmful at high levels. Human and industrial waste contamination of groundwater is increasing, and overuse of groundwater resources has caused water levels to drop, leading to other problems—especially along coastlines. Seawater may move in to replace depleted freshwater, and the ground surface may subside when water is removed from pore spaces in aquifers.
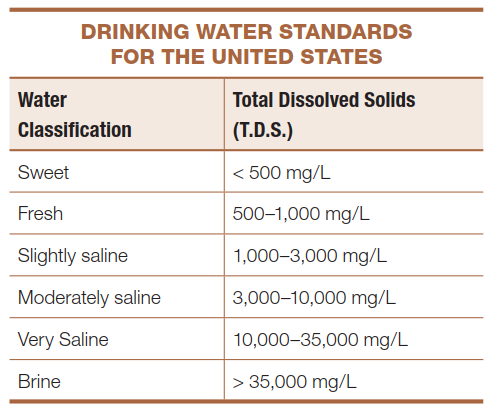
Drinking Water Standards and Total Dissolved Solids. The U.S. Public Health Service has established limits on the concentration of dissolved substances (called total dissolved solids, or TDS) in natural waters used for domestic and other purposes. The table of “Drinking Water Standards for the United States” lists these limits; many other countries, particularly those with chronic water shortages such as those in the Middle East, have much more lenient standards. Sweet water is preferred for domestic use and contains fewer than 500 milligrams (mg) of total dissolved solids per liter (L) of water. Fresh and slightly saline water (TDS of 1,000–3,000 mg/L) is suitable for livestock and irrigation, whereas water with higher TDS is unfit for humans or livestock. Irrigation using high‑TDS water is not recommended, because the water evaporates but leaves dissolved salts and minerals behind, degrading and eventually destroying land productivity.
Either a high amount of total dissolved solids or the introduction of a specific toxic element can reduce groundwater quality or contaminate it. Most TDS in groundwater are salts derived from dissolution of local bedrock or soils derived from bedrock. Salts can also seep into groundwater supplies from the sea along coastlines, particularly if water is being pumped out for use. In such cases, seawater often moves in to replace depleted freshwater—a process known as seawater intrusion or seawater incursion.
Hard Water and Common Dissolved Ions. Dissolved salts in groundwater commonly include bicarbonate (HCO₃⁻) and sulfate (SO₄²⁻) ions, often associated with other ions. Dissolved calcium (Ca²⁺) and magnesium (Mg²⁺) ions can cause water to become “hard.” Hard water is defined as containing more than 120 parts per million of dissolved calcium and magnesium. The dissolved ions in hard water make it difficult to lather soap and form a crusty mineral buildup on faucets and pipes. Adding sodium (Na⁺) in a water softener can soften hard water, but people with heart problems or those on a low‑salt diet should avoid this. Hard water is common in areas where groundwater has moved through limestone or dolostone rocks, which contain high concentrations of Ca²⁺‑ and Mg²⁺‑rich rocks that groundwater easily dissolves.
Human‑Induced Groundwater Contaminants. Groundwater may contain many other contaminants, some natural and others resulting from human activity. Human pollutants include animal and human waste, pesticides, industrial solvents, road salts, petroleum products, and other chemicals—a serious problem in many areas. Some of the biggest and most dangerous sources of groundwater contamination include chemical and gasoline storage tanks, septic systems, landfills, hazardous waste sites, military bases, and the widespread use of road salts and chemicals such as fertilizers or pesticides.
The Environmental Protection Agency (EPA) has led the cleanup of spills from leaking chemical storage tanks in the United States. An estimated 10 million buried chemical storage tanks exist in the U.S., containing gasoline, oil, and hazardous chemicals. These tanks can leak over time, and many older ones have needed replacement in the past two decades, bringing a new generation of tanks that should last longer and corrode less. Home and commercial septic systems pose serious threats to some groundwater systems; most are designed to work effectively, but some were installed improperly or poorly designed. In many cases, groundwater supplies have been contaminated by chemicals poured down drains, entering the septic system and then the groundwater system.
More than 20,000 known and abandoned hazardous waste sites exist in the United States. Some contain many barrels of chemicals and hazardous materials that can and do leak, contaminating water supplies. Landfills may also contain hazardous chemicals; when designed, landfills are supposed to incorporate a protective impermeable bottom layer to prevent chemicals from entering the groundwater system. However, some chemicals erroneously placed in landfills sometimes burn holes in the basal layer, making their way (along with many other chemicals) into the groundwater system.
In parts of the country that freeze, road salts are commonly used to reduce ice on roads. These salts dissolve in rainwater and can eventually make their way down into aquifers, turning an aquifer salty. Together with chemicals from lawn and farm field fertilization and pesticide application, the concentration of these chemicals becomes significant for the safety of underlying water quality.
Residence Time of Groundwater Pollutants. Groundwater contamination, whether natural or human‑induced, is a serious problem because of the importance of the limited water supply. Pollutants in the groundwater system do not simply wash away with the next rain, as many dissolved toxins in surface water systems do. Groundwater pollutants typically have a residence time (average length of time they remain in the system) of hundreds or thousands of years. Many groundwater systems can clean themselves of natural biological contaminants using bacteria, but other chemical contaminants have longer residence times.
Arsenic in Groundwater. In parts of the world, many people have become sick from arsenic dissolved in groundwater. Arsenic poisoning leads to a variety of horrific diseases, including hyperpigmentation (abundance of red freckles), hyperkeratosis (scaly skin lesions), cancerous skin lesions, and squamous cell carcinoma. Arsenic may enter the food chain and body in several ways. In Guizhou Province, China, villagers dry their chili peppers indoors over coal fires; unfortunately, the coal is rich in arsenic (containing up to 35,000 parts per million arsenic), and much of this arsenic transfers to the chili peppers during drying. Thousands of local villagers now suffer arsenic poisoning, with cancers and other forms of the disease ruining families and entire villages.
Most naturally occurring arsenic enters the food chain through drinking contaminated groundwater. Arsenic in groundwater commonly forms by the dissolution of minerals from weathered rocks and soils. In Bangladesh and West Bengal, India, 25–75 million people are at risk for arsenosis because of high concentrations of natural arsenic in groundwater.
Since 1975, the maximum allowable level of arsenic in drinking water in the United States has been 50 parts per billion (ppb). The EPA has considered adopting new standards on allowable arsenic levels. Scientists from the National Academy of Sciences recommend lowering allowable arsenic to 10 ppb, but this level was overruled by the Bush administration. The issue is cost: the EPA estimates that it would cost businesses and taxpayers $181 million per year to bring arsenic levels to the proposed 10 ppb level, although some private foundations suggest this estimate is too low by a factor of three. They estimate the cost would be passed to consumers, and residential water bills would quadruple. The EPA estimates that health benefits from such a lowering would prevent between 7 and 33 deaths from arsenic‑related bladder and lung cancer per year. These issues reflect a delicate and difficult choice for the government. The EPA tries to “maximize health reduction benefits at a cost that is justified by the benefits.” How much should be spent to save 7–33 lives per year? Would the money be better spent elsewhere?
Arsenic is not concentrated evenly in the U.S. groundwater system or anywhere else in the world. The U.S. Geological Survey issued a series of maps in 2000 showing arsenic concentrations in tens of thousands of groundwater wells across the United States. Arsenic is concentrated mostly in the Southwest, with a few peaks elsewhere such as southern Texas, parts of Montana (due to mining operations), and parts of the upper plains states. A remediation plan targeting the highest arsenic concentrations would likely be the most cost‑effective and have the highest health benefit.
Contamination by Sewage. A major problem in groundwater contamination is sewage. If chloroform bacteria (coliform bacteria) enter the groundwater, the aquifer is ruined, and care must be taken with sample analysis before water is used for drinking. In many cases, sand filtering can remove bacteria, and aquifers contaminated by coliform bacteria and other human waste can be cleaned more easily than aquifers contaminated by many other elemental and mineral toxins. Although serious, detailed discussion of groundwater contamination by human waste is beyond the scope of this encyclopedia; readers are referred to sources listed at the end for more detailed accounts.
Seawater Intrusion in Coastal Aquifers. Encroachment of seawater into drinking and irrigation wells is an increasing problem for many coastal communities worldwide. Porous soils and rocks beneath the groundwater table in terrestrial environments are generally saturated with fresh water, whereas porous sediment and rock beneath oceans are saturated with salt water. In coastal environments, a boundary must exist between fresh groundwater and salty groundwater. In some cases this is a vertical boundary; in other cases, the boundary is inclined, with denser salt water lying beneath lighter fresh water. In areas with complex or layered stratigraphy, the boundary may consist of many lenses.
In normal equilibrium, the boundary between fresh and salty water remains rather stationary. During drought, the boundary may move landward or upward; during excessive precipitation, it may move seaward and downward. As sea levels rise, the boundary moves inland, and wells that formerly tapped fresh water begin to tap salt water—called seawater intrusion or encroachment.
Many coastal communities have been highly developed, with residential neighborhoods, cities, and agricultural users obtaining water from groundwater wells. When these wells pump more water out of coastal aquifers than is replenished by new rainfall and other inputs, the fresh water lens resting over the salt water lens is depleted. This causes salt water to move into empty pore spaces, replacing fresh water. Eventually, as pumping continues, the fresh water lens becomes so depleted that wells begin drawing salt water, becoming effectively useless. This is another way salt water intrusion can poison groundwater wells. In severe drought, the process may be natural, but in most cases seawater intrusion is caused by over‑pumping of coastal aquifers, exacerbated by drought conditions.
Many places in the United States have suffered from seawater intrusion. For instance, many East Coast communities have lost use of their wells and had to convert to piped water from distant reservoirs for domestic use. In a more complicated scenario, western Long Island, New York, experienced severe seawater intrusion into its coastal aquifers because of intense overpumping in the late 1800s and early 1900s. Used water once returned to the aquifer by septic systems began to be dumped directly into the sea when sewers were installed in the 1950s, with the result that the water table dropped more than 20 feet over 20 years. This drop was accompanied by additional seawater intrusion. The water table began to recover in the 1970s when much of the area converted to using water pumped from reservoirs in the Catskill Mountains north of New York City.
Summary. Most of the world’s freshwater is locked in glaciers or ice caps, and about 25 percent of freshwater is stored in the groundwater system. Water in the groundwater system is constantly but slowly moving, being recharged by rain and snow that infiltrate the system, and discharging in streams, lakes, springs, and extracted from wells. Water that moves through a porous network forms aquifers; underground layers that restrict flow are known as aquicludes. Fracture zone aquifers comprise generally nonpermeable, nonporous crystalline rock units, but faults and fractures that cut the rock create new or secondary porosity along the fractures. If exposed to the surface, these fractures may become filled with water and serve as excellent water sources in dry regions.
The groundwater system is threatened by pollutants ranging from naturally dissolved but deadly elements such as arsenic, to sewage, to industrial wastes and petroleum products that have leaked from underground storage containers or were carelessly dumped. Some chemical elements have a short residence time in the groundwater system and are effectively cleaned before long, but other elements may last years or thousands of years before the groundwater is drinkable again.
FURTHER READING: Alley, William M., Thomas E. Reilly, and O. L. Franke. Sustainability of Ground-Water Resources. Reston, Va.: United States Geological Survey Circular 1186, 1999.
Ford, D., and P. Williams. Karst Geomorphology and Hydrology. London: Unwin‑Hyman, 1989.
Keller, Edward A. Environmental Geology. 8th ed. Englewood Cliffs, N.J.: Prentice Hall, 2000.
Kusky, T. M. Floods: Hazards of Surface and Groundwater Systems. New York: Facts On File, 2008.
Skinner, Brian J., and Stephen C. Porter. The Dynamic Earth, an Introduction to Physical Geology. 5th ed. New York: John Wiley & Sons, 2004.
Date added: 2026-04-26; views: 7;
