Evolutionary History of Biological Interactions
During most of the twentieth century, researchers assumed a coevolutionary framework for plant-animal interactions. Coevolution occurs if species or groups of species undergo mutual adaptations. Mutual adaptations occur if a change in the traits of one group leads to a change in traits of the other group, which is followed again by a change in the traits of the first group. Coevolution can occur in mutualisms as well as antagonisms and is assumed to have produced much of the Earth’s biodiversity. While adaptations undoubtedly occur—for example, figs dispersed by primarily visually guided birds are typically small, conspicuously coloured and have little scent, while figs dispersed by primarily olfactory guided nocturnal bats are relatively large, predominantly green or yellow and have a strong smell (Lomascolo et al. 2010)—they are often difficult to pinpoint. Even in the example of figs, it is unclear whether coevolution has taken place since the sensory systems of birds and bats may not have evolved in response to the sensory stimuli of figs or, more generally, fruits. A very good system to study coevolution is the resistance among crop plants and their herbivores, which can evolve adaptations to overcome resistance within a decade (Rausher 2001).
While early classical work on coevolution focused mostly on interactions among a single species of plant and a single species of animal, there is now a consensus that the majority of plants interact simultaneously with many animal species and that these interactions are not independent of each other. Multispecies interactions are embedded in, and thus significantly influenced by, the community context (Strauss and Irwin 2004). This realisation has transformed the concept of coevolution into diffuse coevolution among multiple species where interactions with one species can alter the likelihood and strength of interactions with other species. Diffuse coevolution implies that the selective pressures of different species are not independent of each other. This interdependence is found, for example, in the selection for resistance against herbivores in the ivy-leaf morning glory (Ipo- moea hederacea). Significant negative genetic correlations exist between resistance to deer and to generalist insect herbivory (Stinchcombe and Rausher 2001).
In addition, the resistance loci under selection differ according to the composition of the local herbivore community. Finally, selection for deer resistance depends on the presence of insects. In the absence of insects, the deer resistance of ivy-leaf morning glory is effectively neutral. Thus, the sign and strength of selection can be influenced by the composition of the local community of animals and plants. This is important to note because animal communities can fluctuate widely in species composition and relative abundance of species over time and space. An important implication of this concept is, thus, that the fitness effects of plant-animal interactions are spatially and temporally variable, leading to a mosaic of coevolution (Thompson 2005).
Diffuse coevolution is most apparent if different species interact with the same traits. For example, ants that steal nectar but do not pollinate the flowers of the alpine sky pilot (Polemonium viscosum) select for smaller, narrower corollas, whereas bumblebees, the regular pollinator of that species, select for larger, wider corollas (Galen and Cuba 2001). Galen also showed that corolla shape is influenced by the abiotic environment, which again underscores that the net effects of animals on plants will be spatially and temporally variable. How intricately the interactions among different species can be linked is less apparent if distinct animal species interact with distinct plant traits. The interactions of the wild tobacco Nicotiana attenuata with its pollinators and herbivores provide a good example. Typically, wild tobacco flowers at night and is pollinated by hawkmoths, which are also important herbivores for that species. When hawkmoth larvae vigorously attack the leaves, their oral secretions trigger hormonal responses by the plant, which lead to flowers opening during day time (Fig. 19.21).
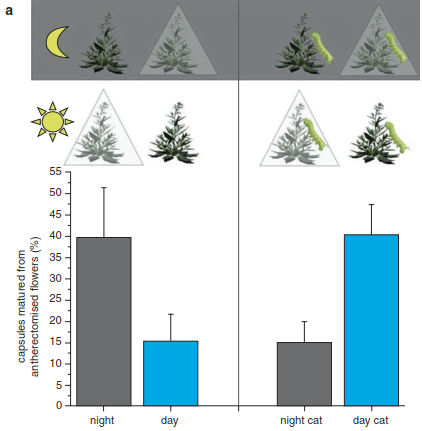
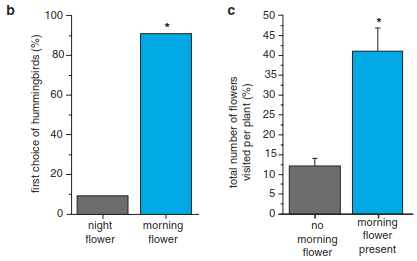
Fig. 19.21. Herbivory influencing pollination system. Herbivory by the larvae of the main pollinator Manduca sexta changed the pollination system in the tobacco plant Nicotiana attenuata. When no herbivores were present, Manduca sexta was the most important pollinator because plants dropped more seeds that were accessible only at night (to Manduca sexta) compared to those that were accessible to pollinators (hummingbirds) only during the day a, left. This pattern was reversed when herbivorous larvae were present on the plants a, right. The shift to hummingbird pollination is explicable by the way plants change to opening flowers in the morning when infested by herbivorous larvae, and hummingbirds prefer to visit flowers that open in the morning b and plants that have flowers in the morning c. (Kessler et al. 2010)
These flowers are then pollinated by hummingbirds, suggesting that wild tobacco changes pollinators to reduce its herbivory load. This study highlights that even seemingly independent interactions, such as those between plants and their pollinators and plants and their herbivores, can be linked by systemic responses by the plant. Consequently, the traditional focus of studying pairwise interactions among plants and animals has shifted towards a more encompassing approach of simultaneously considering multiple interactions.
Expanding the concept of coevolution to diffuse coevolution enables a more encompassing view of plant ecology that has many implications. A first important implication of the concept of diffuse coevolution is that the most apparent animals interacting with plants are not always the most important agents of selection on plants. Microbes and bacteria are ubiquitous selective agents whose effects on plants are often overlooked but widespread. For example, while pollinators and seed dispersers are the animals most evidently interacting with flowers and fruits, respectively, their interactions with plants cannot explain why resources that are made to be eaten, such as nectar and fruits, can be toxic to the extent that they are rarely consumed. For example, deterrent nectar has been found in at least 21 plant families (Adler 2000).
Typically, combinations of chemical repellents and attractants will help reproductive organs to avoid predators while attracting animals’ dispersing genes. The second implication is thus that the effects of all organisms interacting with a given plant trait must be considered in order to understand the mechanisms that structure interactions. The third implication is the lack of reciprocity that is necessarily found in strict coevolution among one plant population and one animal population. The fourth implication is that the results of studies can rarely be generalised across space and time, simply because fluctuations in the biotic and abiotic environment can alter how plants interact with a given species.
An alternative framework to coevolution is ecological fitting. Ecological fitting describes interactions among partners without mutual adaptations and is an appropriate null hypothesis for the concept of diffuse coevolution. Ecological fitting occurs if species interact with those species in a community that match best their own traits without a shared evolutionary history (Janzen 1985). Importantly, however, the concept of ecological fitting can be difficult to disentangle from diffuse coevolution because historical information on coexistence and mutual adaptations is rarely available (except for invasive species). The key difference is that coevolution expects
| 0.2 |
Although ecological fitting is an inevitable and probably frequent process in nature that results from the interaction between highly flexible organisms and highly variable biotic and abiotic environments, it is not easily recognisable. Ecological fitting is thus perhaps most easily recognised in communities with frequent species turnover. Often the species assemblage within communities is not stable, whether owing to climatic changes such as glaciation cycles or to current human-induced changes to the environment such as grazing regimes of livestock. A context in which ecological fitting has attracted a lot of attention recently is that of invasive species, which obviously integrate into communities without sharing a coevolutionary history with the native species. For example, Impatiens glan- dulifera is an Asian invader in Central Europe reducing the pollination success of native species by attracting pollinators with its rich nectar resources (Chittka and Schurkens 2001). More generally, it is hypothesised that phenotypic plasticity is an important prerequisite for ecological fitting and for establishing new interactions with other species (Agosta and Klemens 2008).
Independent of whether or not the interactions among plants and animals instill evolutionary change, it is important to note that plants typically interact not only with a multitude of herbivores, pollinators and seed dispersers but also with bacteria and fungi. Even though bacteria and fungi typically outnumber herbivores and pollinators, the consequences of their interactions with plants are often not fully understood. The pungent taste of chilli (Capsicum spp.) is a good example. In Capsicum chacoense in south-eastern Bolivia there is a strong variability in pungency within and among populations. Interestingly, this variability is directly linked to variations in the damage caused by the fungal pathogen Fusarium semitectum that attacks the seeds. This fungus is the primary cause of seed mortality in C. chacoense, but the likelihood of infection is contingent upon secondary plant constituents. Fungal infection was twice as high in seeds from non-pungent fruits compared to pungent fruits (Fig. 19.22) (Tewksbury et al. 2008). Hemipterans foraging on the fruits facilitate the entry of the fungi into the fruit and its spread between fruits. Most notably, the foraging pressure of Hemipterans was a strong predictor of the proportion of pungent plants at a given site. Taken together, these results suggest that pungency in chillies is an adaptive response to the likelihood of fungal attack. More importantly, it is an example of the use of biochemistry to mediate the interactions with animals and pathogens.
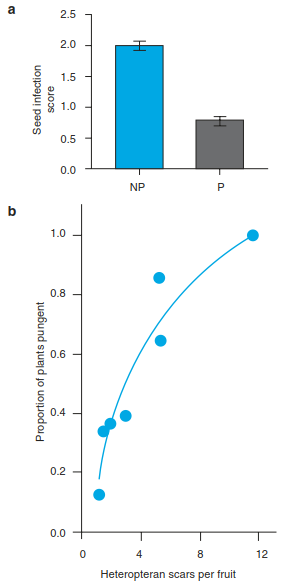
Fig. 19.22. Plant–herbivore–fungi interactions. Capsicum chacoense is naturally polymorphic for the production of pungent capsaicin. a Non-pungent seeds (NP) are twice as likely to be colonised by fungi (Fusarium semitectum) as pungent seeds (P). b The proportion of plants producing capsaicin within populations is a function of the predation pressures by Hemiptera, which puncture fruit skin and thereby facilitate the infection by the fungi (Tewksbury et al. 2008)
Biochemistry is among the main mechanism by which plants structure interactions with their environment as their immobility precludes their seeking out favourable environmental conditions. Plants’ biochemicals can be either very specific, targeting only one group or even species (see volatile signalling in what follows), or relatively general, affecting multiple groups of species. An important reason to expect general effects is that many biochemical pathways in plants produce a variety of end products that in turn can affect a variety of animals. Again, pungency in chilli illustrates broad as well as specific effects. Pungency does not only defend the chilli plant against fungi attack but also against seed predation by mammals which is reduced in pungent seeds (Tewksbury et al. 2008). In contrast, birds, the legitimate seed dispersers of chilli plants, are unaffected by the presence of capsaicin, the substance that gives chilli its pungent taste. Thus, capsaicin has general deterrent effects on both mammals and fungi but does not affect birds.
The fact that plants mediate their interactions through biochemistry can lead to the deceiving impression that plants are relatively inactive compared to animals. Yet this is not the case, as we will see in the following sections. Plants are active players that can quickly increase their defences in response to attacks by herbivores; they attract the enemies of their herbivores and use “signals” to increase their visibility and memorability for the animals they depend on.
Plants possess a very diverse array of secondary compounds with far more than 200,000 substances known so far and many more being discovered each year. The term secondary compounds encompasses all metabolic products without a known function in the primary metabolism of plants. In other words, all substances that are not known to be involved in the vital functions of growth, development, maintenance and reproduction of plants are included under this umbrella term. While the functions of the majority of secondary compounds are not known, a large number of them contribute to mediate plant-animal interactions. In general, three classes of substances are involved: pigments that impart colour to flowers and fruits, volatiles that attract pollinators but also recruit the enemies of herbivores, and deterrent substances that alter the quality of food.
Plant-Herbivore Interactions: Defenses & Coevolution
Herbivores consume large proportions of the biomass of plants in most ecosystems. It is thus not surprising that herbivores can alter the composition of plant communities. Herbivory also forms the major process through which energy enters the food chain and is of major economic importance as it causes huge economic losses in plant crops cultivated for human consumption, for example, when swarms of locusts plague farmers or by post-harvest losses.
However, herbivory represents a very complex field of interactions between plants and animals. Plants are not just eaten. Herbivores influence plants in many ways, affecting their fitness and growth, as well as the composition, diversity, structure and dynamics of plant communities. Some results of grazing and browsing in plant communities are shown in Fig. 19.23.

Fig. 19.23. Fodder production, grazing and browsing may have very different consequences for plant communities. a In the Rif Mountains in northern Morocco, branches of summer green oaks (Quercus faginea) are cut when animal fodder is in short supply. The trees appear stunted. b Extreme browsing pressure on Quercus coccifera in Jordan leads to sharper and stronger thorns on the small oak bushes. c Strong grazing pressure can also lead to increased frequency of toxic species, for example, Asphodelus div. spec. shown here in pastures of the Plateau Central in Morocco. d If herbs and grasses have already been lost due to excessive grazing, the remaining trees, for example, Argania spinose, are browsed by goats in the Sousse plain in southern Morocco. (Photos: K. Müller-Hohenstein)
Globally, the most important herbivores are the “wild” large vertebrates, such as African and Eurasian ungulates grazing in semiarid tropical regions. Domestic grazers, such as cows and sheep, prevail on artificial pastures in temperate climates. There are also many herbivorous invertebrate species which consume leaves, and also other plant resources; they suck sap, eat pollen or seeds, buds or young shoots. Insects in all stages of development are the most important group of herbivorous invertebrates. They are more specialised than vertebrates (i.e. are mono- or oligophagous), while vertebrates are mostly polyphagous. A special case of herbivory with enormous economic impact is granivory.
Granivores feed mainly or even exclusively on seeds. Only recently has more attention been paid to root feeders (Stein et al. 2010). There is evidence that below-ground herbivory may sometimes be more important in its quantity and effects on plant communities than above-ground herbivory.
Extreme grazing generally leads to the loss of palatable species in plant communities, while especially poisonous and thorny species may expand their population sizes. However, herbivores can also promote plant species richness in many plant communities with medium grazing pressure by reducing the dominance of otherwise highly competitive plant species. The manifold consequences of herbivory depend also on the seasonal development of the different plant tissues. Especially in arid regions, early grazing of not yet fully developed plants has a significant influence on plant survival and fitness.
Two major hypotheses have been formulated concerning plant-herbivore relations: the plant stress hypothesis proposes that physiologically stressed plants become more susceptible to herbivores (Louda and Collinge 1992), while the plant vigour hypothesis postulates that herbivores prefer plants growing vigorously (Price 1991).
Plants use two strategies to cope with herbivores, tolerance and resistance. Resistance is the reduction of the amount of damage caused by herbivores, whereas tolerance describes the reduction of the impact of herbivores on plant reproduction, for example, through rapid regrowth or increased photosynthetic activity. Resistance can be caused by biochemical and mechanical traits; the latter include thorns, latex, trichomes and other structures that impede browsing.
A fundamental principle is that both tolerance and resistance are costly to plants but that these costs are balanced through an increase in growth, reproduction and survival (Fig. 19.24). The costs of both strategies can also explain why there is a large interspecific difference among plants in their levels of resistance and tolerance. Costs are opportunity costs, for example, of resources devoted to defences or regrowth that could otherwise be used for reproduction. Biochemical defences also include the potential costs of deterring those animals plants want to attract, such as seed dispersers and pollinators, if the biochemicals are broadly deterrent and not restricted to the tissue vulnerable only to herbivores.
Fig. 19.24. Effects of herbivory on plant growth and fitness. Cases A and B assume that there is a linear relation between the intensity with which a plant is eaten and fitness (e.g. seed yield per plant). In case C herbivore pressure without visible effects is initially tolerated; only later do significant losses occur. In cases D and E grazing stimulates at first, stimulation may lead to increased fitness E, after the initial transient increase there may also be a decrease D. (After McNaughton 1983)
Plant defences can be classified according to their specificity. Most plants possess an array of general, broadly deterrent defences against herbivores such as tannins, for example. These defences are typically not very strong and can be overcome by herbivores. Thus, in addition to those broadly active defences, many plants produce more potent, specific defences that act against specialist herbivores. It is here where an arms race can occur between plants and their herbivores. Arms races are characterised by three steps. First, plants develop secondary compounds by which they defend themselves against herbivores. Plants producing biologically active compounds gain fitness benefits by reducing the numbers of herbivores.
Second, as those defences reduce the fitness of herbivores, there is often strong selection on herbivores to overcome plant defences. This step is facilitated by two factors, the significantly shorter generation times of herbivores and pathogens (compared to most plants) and the higher population density of most antagonists. Third, plants develop new secondary compounds to fend off the antagonists that developed resistance to the previously evolved substances. A given plant still retains the original secondary compound because not all of its enemies will have evolved to overcome it. Thus, within-plant diversity of secondary compounds is driven, at least in part, by the ecological diversity of the enemies a plant may face and escalating arms races with some of these enemies. It is easy to see that arms races are a form of coevolution. Interestingly, such arms races of mutual counteradaptations are not found if plants employ a strategy of tolerance to cope with herbivores because tolerance does not reduce the fitness of herbivores, so it does not lead to counteradaptations by them.
Another way of classifying plant defences is according to their presence in plant tissue. Constitutive defences are continuously present, whereas induced defences are produced by a plant only if it is attacked by herbivores or if another plant in its direct vicinity is attacked. Induced defences are thus one way for plants to reduce the costs of defending themselves since they incur them only when the defences are actually needed. This is particularly so because attacks by herbivores are unpredictable in space and time. Notably, a plant relying solely on induced defences to fend off herbivores may be at a selective disadvantage relative to plants relying also on constitutive defences because there is a time lag before induced defences can be mobilised.
This time lag can be reduced if plants prepare for the mobilisation of defences, a process known as priming, which refers to the synthesis of precursors of induced defences so that the time lag is reduced. For example, plants can use basic building blocks in the phenolpropanoid pathway such as chalcone to synthesise defensive compounds such as tannins. Indeed, priming can be induced by volatiles released from other plants that are associated with herbivore attack (Heil and Karban 2010). Another area in which priming is important is when a plant’s reaction to one stressor enables a faster or stronger response to a second stressor. This often occurs if the biochemicals used in response to the two different stressors are derived from the same biosynthetic pathway because the same precursors can be used for both types of biochemicals. A good example of such a pathway is, again, the phenylpropane pathway in plants from which diverse substances originate from anthocyanins as plant pigments to various defensive compounds such as tannins and flavones (Schaefer and Rolshausen 2006).
Induced defences thus require that a plant senses that an attack by herbivores is likely to occur. This occurs obviously if the plant itself is attacked. Induced defence may result from the systemic transport of defensive metabolites from the area under attack to other parts of the same plant. As such it could be an unavoidable but slow process. Yet, the alert of an attack can also be transmitted through new substances produced after the attack has occurred. This chemical signal can be transmitted through the vascular system of the plant, or the signal can involve the release of volatile chemicals into the air from affected leaves of the plant and their detection by other parts or be transported through the networks of roots. How plants perceive airborne signals remains unknown. What is known, though, is that plants can adsorb and rerelease volatiles from neighbouring plants (Himanen et al. 2010).
Airborne signalling can be more rapid than vascular signalling, particularly in neighbouring leaves from different branches. It is easy to understand how an airborne signalling system can evolve since herbivory entails the destruction of cells with an unavoidable emission of substances into the air. Volatiles may thus be the unavoidable consequence of tissue destruction by herbivores, or they may constitute alerting signals specifically evolved to transmit information. Several factors, such as wind, temperature and nutrient availability, are known to influence the emission of volatiles. Distinguishing whether volatiles are necessary consequences of tissue destruction or evolved signals will allow us to understand whether plants evolved a communication system of alerting signals or whether they obtain information from cues, which are unavoidable by-products of herbivory.
Given that volatiles can indicate an attack of herbivores, plants may also be able to “sense” the attack on a neighbouring plant. One classical study (Karban et al. 2000) demonstrated that wild tobacco (Nicotiana attenuata) growing in a field in 0-15 cm distance to experimentally- clipped sagebush (Artemisia tridentata) became more resistant to herbivores than control plants near to unclipped sagebush (Fig. 19.25). The volatile signal inducing resistance in wild tobacco was an epimer of methyl jasmonate. Undamaged sagebrush individuals near clipped conspecif- ics also experienced reduced herbivore damage relative to controls. Although, in general, odours can travel large distances, distances recorded in experiments were typically only in the range of 10-15 cm. Thus, communication among plants through induced volatiles seems to be restricted to short distances. Such short distances are typical in grasslands but unlikely to occur commonly among trees in forests because the effects of competition among neighbouring plants are likely to outweigh the benefits of interchanging alerting signals. Hence, the topic of alerting signals between different plant individuals has not been fully resolved.
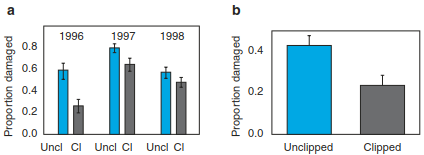
Fig. 19.25. Communication between plants through volatiles induces resistance to herbivores. a Maximum proportion of leaves damaged by grasshoppers during three seasons on tobacco plants near clipped (Cl) or unclipped (Uncl) sagebrush (means ± 1 SE). b Maximum proportion of leaves damaged by cutworms during 1996 season on tobacco plants near clipped or unclipped sagebrush (means ± 1 SE). (Karban et al. 2000)
Apart from indicating the likelihood of an imminent attack by herbivores, induced volatiles can also have other functions: they can deter herbivores directly or attract their enemies, both predators and parasites. That plants can attract predators and parasites of herbivores underscores again the point that plants are active players in their interactions with other organisms. While there had been a debate on the effectiveness of volatiles under natural conditions, a few studies have shown a twofold reduction in the rate of oviposition by other herbivores and a fivefold increase in the rate of predation on eggs in response to induced volatiles. These numbers correspond to 90% reduction in herbivore load through the use of induced volatiles (Kessler and Baldwin 2001), suggesting that communication to the enemies of their enemies can be strongly beneficial for plants.
There is evidence that plants actively modify their olfactory communication in response to herbivory attack. Herbivore-damaged tobacco plants release volatile blends during the day that attract parasitoids of their herbivores, whereas those released at night repel herbivores that forage during the night. This evidence suggests that plants have indeed evolved an active SOS communication to fend off herbivores. Given that many predators and parasites of herbivores are insects such as wasps that search for their prey using volatiles, it is easy to envision how plants’ SOS communication system evolved as those predators are already pre-adapted to react to odours.
Another intriguing aspect is that some plant species have evolved mutualisms with other animals, often ants, as a protection against her- bivory. These plants provide ants with food and with housing (domatia) in the form of hollow swellings. The ants, in return, protect their host plant from herbivores and sometimes also from competitors. In addition, plants can take up nutrients from the decomposing detritus transported into the plants by the ants, as well as from the CO2 respired by the ants (Treseder et al. 1995).
Interestingly, as in most mutualisms, the effects of plants and ants on their partners can vary. While some interactions evolved to the state of obligate mutualisms where ants are truly symbionts living only on those plants, other ant species destroy the flowers of their hosts to induce them to invest in growth rather than reproduction as these ants benefit more from plant growth. Again, plant chemistry can play an important role in structuring these ant-plant mutualisms. In exceptional cases, ants can only digest the type of carbohydrates produced by their host plant, effectively limiting their options to thrive on other plant species.
Another potential form of protection against herbivores is mimicry. Mimicry describes the adaptive resemblance between a model species and a mimic. Mimicry occurs if the mimic gains adaptive benefits by duping animals into confusing it with the model (Fig. 19.26). This confusion between mimic and model is the central element in mimicry and distinguishes it from related phenomena such as the exploitation of innate sensory biases of perceivers, which arise through their sensory system. Pollinators, for example, can have innate biases for visiting large floral displays (Naug and Arathi 2007).

Fig. 19.26. Mimicry as a protection against herbivory. Seeds of Margaritaria nobilis are brightly coloured, like fruits, but offer no reward to seed dispersers in terms of fruit pulp
Mimicry may occur interspecifically if individuals from a poorly defended species resemble individuals from a better defended species and, thus, gain protection from herbivores. This is a long-standing hypothesis that may pertain to mistletoes and vines that might benefit from being mistaken as host plants. One of the main challenges has been to show an adaptive benefit of such a resemblance. A recent study on the Neotropical vine Boquila trifoliolata is consistent with the mimicry hypothesis by showing (1) that leaves of this species resemble the foliage of the tree B. trifoliolata it is climbing and (2) that leaf herbivory is greater on vines climbing leafless trunks compared to those climbing trees with foliage (Gianoli and Carrasco-Urra 2014).
Over the last 15 years a hotly debated question has been whether plants use leaf colour as a visual signal to communicate their defensive strength to potential herbivores. The idea was originally proposed to explain the exuberant yellow and red leaf colours during fall in the Northern Hemisphere (Archetti 2000; Hamilton and Brown 2001). The basic reasoning behind this hypothesis is that plants could benefit from such a signal of strength by reducing the attack rate of herbivores, while herbivores could benefit from such a signal by finding the least defended plants more quickly.
While studies were quick to show a correlation between non-green leaf colours and herbivore attack rates (e.g. Hagen et al. 2003), it proved more difficult to establish that herbivores respond to leaf colour (and not to an associated trait) and that this association represents actually a signal that evolved for the effect of communicating to herbivores. Arguably, the best experimental evidence stems from the Southern Hemisphere. Leaves of the shrub Pseudowintera colorata vary in colour from green to red. Leaves with wider red margins contained higher concentrations of the secondary compound polygodial and experienced less herbivory. Crucially, experiments demonstrated that larvae of the generalist herbivore Ctenopseustis obliquana responded to the colouration of leaves when they avoided red leaves (Cooney et al. 2012). This study suggests that plants can use aposematic warning colours to communicate to herbivores and that herbivores attend to variation in leaf colouration.
Date added: 2026-04-26; views: 1;
