Competition and Its Consequences for Plant Community Structure and Diversity
So far, this chapter has largely focused on pairwise interactions, where one species is (temporarily) superior to another. In a community setting, many species are simultaneously competing with each other, typically more so the nearer they are to each other. The consequences of plant competition for plant communities as a whole are much less straightforward. The key problem is the existence of competitive loops, where species A, B and C compete for several resources. For any resource alone, we may find a competitive hierarchy (e.g. for nitrogen: A > B > C), but that hierarchy may be different for another resource (e.g. for light: B > C > A). In such circumstances, the dominance of C over A for light might actually benefit B in its nitrogen competition. This situation has been called “my enemy’s enemy is my friend”, and it leads to a rather unpredictable outcome of competition for multiple resources. In fact, since experimental effort increases dramatically with the number of species considered, competition is often invoked in community experiments, rather than being directly proven. What can be observed is a decrease in the biomass of a target species, called a phytometer, for example, as diversity increases or soils become more fertile. This is attributed to greater resource depletion due to competition. Thus, competition is typically inferred as a driving force, either conceptually or mathematically.
Certain concepts link competition to abundance patterns in vegetation. For example, Keddy (1989) suggested the idea of competitive hierarchies, where a superior species is able to exclude all others, the second-most dominant all those below it and so forth. This would lead to a zonation pattern, with the most dominant species occupying the best growth conditions, although all species would grow best there. Experimental evidence dates back to the legendary Hohenheimer Grundwasserversuch of Ellen- berg and Walter in 1952 (Box 19.3). It shows that the individual niche optimum (the “autecologi- cal optimum”) will differ from a plant’s position in the vegetation, surrounded by other species (the “synecological optimum”, although optimum is arguably the wrong word here). However, not all experimental evidence, of which there is little, agrees.
As competition changes species composition, transitions from one kind of plant community to another occur, altering the environment in the process (succession). In some systems these alterations are cyclical, for example, because the new community is more flammable and fire will burn the vegetation down to the ground, restarting a succession. At the landscape level, several different stages of this cycle are present, so species coexist, not in a patch, but at a higher spatial scale. The resulting dynamic mosaic of succes- sional stages has been observed in heathlands and rain forests (the “mosaic-cycle concept” of Remmert 1991). As this concept is phenomenological (i.e. describes the outcome rather than the mechanisms), it is more of historic interest and adds little to our understanding of competitive interactions in plant communities.
Competition in Concert with Trophic Interactions.In any given plant community, competition is not the only biotic interaction at work. In fact, a rich literature investigates the effect of other interactions on the importance of competition. Livestock grazing, as well as the more selective browsing of wood plants by ungulates, is the most commonly investigated form of biotic process interfering with plant competition. The field evidence is unequivocal: grazing suppresses the dominant species, increases light availability, creates open sites for germination, removes nutrients from the system and, overall, leads to or maintains higher species richness. The main reason for the overall positive effect of grazing is that dominant species lose more biomass, in absolute terms, than rare ones. Thus, grazing, as well as other frequency- dependent damage such as hemiparasitism, often acts as an equalising mechanism. Browsing effects in contrast are highly specific to the herbivore and its level of foraging preferences. An indiscriminate browser suppresses the dominant species, similar to grazing, but strongly discriminating browsers (or grazers) can have effects similar to those of plant pathogens.
Less obvious, but probably even more common, is the effect of pathogens, in particular fungal infections and herbivory by caterpillars, weevils and suchlike, on plant communities. In contrast to grazers, these pathogens are typically highly specialised and may locally exterminate their host. Still, pathogens can contribute to the dynamic long-term coexistence of plant species by suppressing the most abundant species. A key feature is that pathogen transmission is density dependent, so that as a species becomes rarer in the community, it is less often attacked by pathogens. Hence, pathogens lead to the stabilising condition required for coexistence among competing species (Janzen-Connell hypothesis, initially proposed particularly for tropical tree seedlings) (Fig. 19.14). The mechanism resembles the effect of competition, that is, one species dominating another, but the actual agent is not the dominating plant, but the pathogen that suppresses the subdominant. The term apparent competition describes this situation aptly: it looks like competition but is really the outcome of selective suppression by a higher trophic level. From a theoretical point of view, herbivory can be seen as competition for attack-free space and has been shown to be mathematically equivalent (Chesson and Kuang 2008).
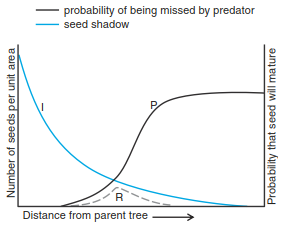
Fig. 19.14. The Janzen–Connell hypothesis. Shown is the recruitment curve R as the result of two distance-dependent processes: the number of seeds decreases with distance to the parent tree (I), while the chance of escaping predation by a specialist seed predator (P) increases. Modified from Janzen (1970), but independently derived also by Connell (1971)
Similar to the ambivalent effect of facilitation (which may shift from a beneficial to a competitive relationship), “pathogens” (fungi, endophytic algae) may also occasionally have positive effects on host plants. Fungal endophytes spend their lives in the tissue of commercially important grasses, increasing or decreasing their transpiration, reducing their palatability to grazers and sometimes increasing their growth rate. This relationship progresses, depending on the plant species, from antagonistic to commensalistic to symbiotic. Accordingly, the way pathogens affect plant community composition and diversity is not easy to anticipate.
Consistent Determinants of Plant Community Structure and Diversity.Predicting the effects of competition on community structure is, in principle, difficult: all species interact in slightly different ways, and these interactions vary through time. Additionally, the outcome of this confusion of interactions should change with resource availability, depend on the sequence of establishments (founder effect) and be susceptible to disturbances and management. In reality, however, plant communities are far more predictable than the variety of possible interactions would suggest. There are several reasons:
1. Often one environmental factor overrides all the biotic interaction details. This is obvious in stressful environments, such as deserts, polar regions or salt marshes. It is, however, also true in dark forest understoreys, waterlogged soils or highly grazed pastures. Only species able to cope with such constraints will be able to survive, and we find only few alternative survival strategies for each situation (e.g. succulence or seed banks in the case of deserts; thorns, toxins or growth meristems close to the ground in the case of grazing). In a simple sentence: competition may be intense, but it is unimportant relative to other factors.
2. Evolution cannot combine growth strategies and plant traits freely. New species come with the baggage of a phylogenetic past, and they are modifications of existing species, not optimally assembled from whatever Nature has to offer. Hence, many plant families have never managed to become established in regions beyond their evolutionary origin, have never been able to grow rapidly or develop a tolerance to drought. Crassulaceae, for example, have developed a rare photosynthetic trick (C4-CAM, Sect. 12.1 in Chap. 12), but they are void of secondary growth (Sect. 12.5, Chap. 12) and hence do not grow into trees. As a consequence of the phylogenetic conservatism in trait evolution, we find specific families or even genera dominating specific systems (e.g. Dipterocarpaceae in the Asian tropics, Poaceae in temperate grasslands, Cactaceae/Euphorbiaceae in deserts).
3. Plasticity within species leads to local acclimation (phenotypic plasticity) and allows a dominant species to be dominant over a large range of environmental conditions (including local adaptations due to genotypic plasticity, e.g. ecotypes). Temperate European forests are dominated, in their natural state, by European beech Fagus sylvatica over a wide range of soil and climatic conditions (Fig. 19.4). Similarly, couch grass Elymus repens is a pioneer grass species worldwide, in nutrient-rich arid systems as much as fertile salt marshes, on sand as much as on loamy soils. Both species display phenotypic plasticity, as well as high genetic diversity across their distributional range and adaptive polyploidy.
4. During successional developments (be it primary succession of glacial deposits, landslides or volcanic rock, or secondary succession following logging or abandonment of arable land), environmental constraints alternate. We can imagine a glacial foreland to be primarily nutrient limited, favouring species with symbiotic nitrogen fixers. Such early pioneers are soon replaced by larger forbs, then shrubs. In due course, as an organic soil layer builds up, nutrient limitations are replaced by competition for light, which is won by trees. Succession is thus a relative predictable sequence of traits and species (Sect. 17.2, Chap. 17).
5. Most species are rare. The reasons are manifold, but point 2 above (limited genetic variability within any given clade) is certainly one of the main culprits. Over centuries and millennia, dominance patterns of plants remain surprisingly stable, as witnessed in pollen records. Despite thousands of plant species in the continental species pool, only the same few handful dominate the pollen record. Abundance is thus not a mere fluke or outcome of ecologic drift, as Hubbell’s neutral theory suggests (Hubbell 2001). Whatever trait combination makes these few species superior, the common species are apparently able to hold on to their dominance and are rarely replaced by (native or non-native) invading species. When this does happen, it can be spectacular (such as the red chinona Cinchona pubescens on the Galapagos Islands or Japanese knotweed Fallopia japonica in parts of Europe). Still, species-induced system changes are rare, and most often human land and pest management are behind observed dramatic changes in species abundances (Sect. 17.2, Chap. 17: human impacts and climate change).
Date added: 2026-04-26; views: 1;
