Cation Fluxes in Terrestrial Ecosystems
Cation supply (in particular of K+, Mg2+, Ca2+) occurs mainly from chemical weathering of primary minerals (Chap. 11) or via dust particles and sea spray entering the ecosystem from the atmosphere. Examples of dust inputs are the formation of loess in the post-glacial period (Blume et al. 2010), the supply of dust from the Sahara to the Amazon delta (Worobiec et al. 2007) and buffering of sulphur-containing emissions by industrial dusts in the 1960s, which delayed acidification of soils, as the ionic charge of deposited material was neutral. The deposition of sea salts from sea spray in coastal areas, that is, sea salt aerosols formed from the ocean, results in strong gradients of Ca2+, Na+ and Mg2+, as well as Cl- and SO42- ions deposited on plant foliage to interior areas (Gustafsson and Franzen 2000).
Cations are taken up by roots and incorporated into plant tissues, where they remain for months to decades depending on foliage lifetimes and decomposition rates (Sect. 16.2.2). However, the return of cations into the soil occurs not only via decomposition but also through leaching from the canopy. Canopy leaching is a consequence of ammonium uptake from atmospheric pollution during which cations are leached buffering the input of protons. Thus, cation concentrations in throughfall are often larger than in bulk precipitation (Sect. 16.1). The accumulation of cations in the litter layer and in soil organic material and dense root systems in the top soil horizons enable direct resupply of cations from decomposing organic material to plants, particularly under nutrient-limited conditions (e.g. tropical and boreal systems). However, cations released from organic matter can also be leached from the soil together with DOC, for example, organic acids. As a consequence, cation concentrations (also aluminium and iron) in the soil solution of upper soil layers decrease and bleached horizons are formed, especially in nutrient-poor, sandy soils (eluvial E horizons). Alkaline saturation increases only in deeper soil layers (B and C horizons).
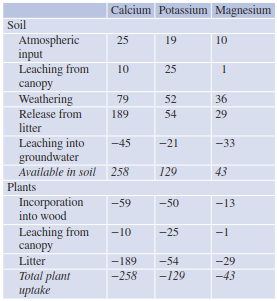
Table 16.7. Cation balance in a spruce forest (in mmol m−2 year−1)
The dynamics from uptake to release are very different for individual elements. In a spruce stand on granite, the calcium fluxes were about twice those of potassium and exceeded those of magnesium five-fold (Horn et al. 1989). As leaching into groundwater occurs for all elements at about the same magnitude, different amounts of Ca2+, K+ and Mg2+ must be supplied via the weathering of primary minerals in the soil profile (Table 16.7). Since weathering of granite is slow, this leads to decreased soil pH and to low forest productivity on acidic bedrock.
Ecosystems cannot avoid the loss of cations. Even in ecosystems without management and with a high root density, cations released during decomposition can be leached with organic acids into deeper layers or downslope. This process is known from the boreal zone as podzolisation (Blume et al. 2010; Weil and Brady 2009) and occurred in Scandinavia, for example, after the land rose (after glaciation) and was evident after only 400 years (Starr 1991). Leaching of cations from an ecosystem into groundwater and lateral transport into other ecosystems may have far- reaching consequences for nitrogen cycles of “supplier” and “receiver” systems. Two interactions are possible (Fig. 16.24).
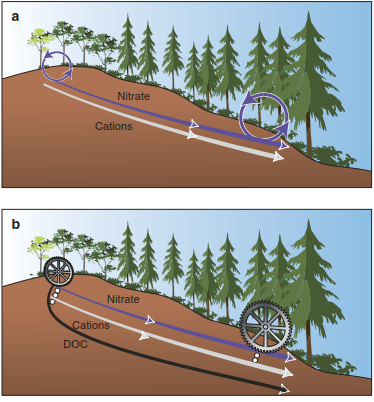
Fig. 16.24. Schematic representation of nitrate and cation fluxes downslope resulting from groundwater run-off in a boreal forest in Scandinavia. Two different mechanisms could explain the high productivity of trees downslope. a High nitrate leaching and associated cation loss that accumulate downslope and increase resource availability for growth downslope. b Low nitrate loss but large cation loss in association with DOC. (After Hogberg 2001)
(A) Nitrate (or sulphate) is leached and carries cations away, leading to an increased total turnover; this is the “classic” assumption. (B) DOC is leached and cations accumulate in deeper soil horizons. As DOC is microbiologically mineralised during transport, there is secondary cation accumulation, leading to higher pH values deeper in the soil profile or downslope. Thus, nitrogen mineralisation also increases, allowing for high tree productivity (mechanism B in Fig. 16.24). The transport of DOC appears to dominate in the boreal climate (Hogberg 2001) on acid substrates (granite), effecting relocation of cations, for example, on a slope (Guggenberger and Zech 1993). It is often assumed that other vegetation types, particularly in the tropics, are so well adapted to poor nutrient conditions that cation losses can be ignored. However, Chadwick et al. (1999) have shown that cation losses also occur in tropical climates, that is, for vegetation on lava flows of different ages (Fig. 16.25). As soon as weathering rates decline, the ecosystems are supported by the atmospheric deposition of cations from Central Asia more than 6000 km away.
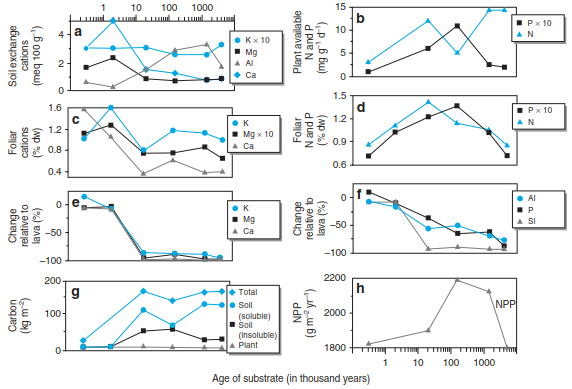
Fig. 16.25. a–d Changes in K, Mg, Al, Ca, P, and N concentrations in soils derived from weathered lava and in leaves along an age gradient on Hawaii. e, f The relative changes in soil nutrients compared to lava and g carbon pools as well as h NPP are also shown. (Vitousek et al. 1997; Chadwick et al. 1999)
Natural processes causing cation loss may be accelerated by strong acids, particularly if they form acid anions that then enter the groundwater. Sulphate, but also chloride and nitrate, belongs to these acid anions, insofar as they are not used in lower soil layers and, thus, enter the groundwater. Such strong acids do not occur in excess under natural conditions. However, since industrialisation, ecosystems have been increasingly impacted by the atmospheric deposition of acids. In Europe, this has caused soil acidification over a period of about 30 years (Schulze 1989), with base saturation in all soil horizons decreasing from 10 to 50% at the start of the period to 5% on average a few decades later (Ulrich 1987). In Sweden, some soils even lost 70% of their exchangeable base cations in the mineral soil, but not in the organic layer (Hogberg et al. 2006).
Forest vegetation should be adapted to soil acidification because under natural conditions, trees occupy sites across a very wide range of soil acidity (Ellenberg 1978). The phenomenon of forest decline in Central Europe prompted a controversy about whether:
- The observed loss of needles and discolouration were natural and would have occurred without the atmospheric input of acids.
- The damage was triggered by organisms (pests and pathogens).
- The damage was a direct response to atmospheric pollutants or a consequence of acidification of soils.
Schulze and Lange (1990) argued that the different paths leading to damage, decline and, eventually, death of trees did not need to be exclusive but become effective at different times over time (Fig. 16.26).
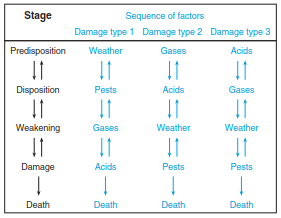
Fig. 16.26. Conceptual framework of a combination of factors leading to forest decline (Schulze and Lange 1990)
To evaluate this framework, it was necessary to examine the consequences of a wide range of possible factors and to identify the main cause(s):
- The primary impact of pests and pathogens could not be demonstrated, although several insects and pathogens (bark beetles, stem rot) attack weakened trees and, ultimately, cause their death.
- During the twentieth century, high concentrations of pollutant gases, for example, SO2 and NOx, in emissions from industries caused damage, but the more recent damage symptoms were different. These new symptoms were related to tropospheric ozone, but the effect of ozone was shown to be rather complex and not straightforward (Chap. 5).
- Acidification of soils causes root damage, particularly due to the release of mono-nuclear aluminium (Al) species at low pH values (increased solubilisation of Al oxides and Al silicates at pH < 5; Sect. 11.5 in Chap. 11). However, individual tree species react differently to Al. Furthermore, Al also reacts with phosphate in the mycorrhiza and is immobilised (Kottke and Oberwinkler 1986). Thus, soil acidification alone could not explain the observed damage.
- One type of forest damage, characterised by yellowing of needles caused by magnesium deficiency, could be explained as a consequence of pollutants at the ecosystem scale (Schulze 1989). Soil acidification strongly reduces the availability of magnesium (and calcium) to plants. This is not only caused by the reduced base saturation of the soil exchanger occurring simultaneously with acidification, but also by competitive inhibi
tion of Mg uptake by ammonium. In addition to ammonification, ammonium in the soil originates from atmospheric deposition, particularly from animal husbandry. Ammonium causes the release of Mg from exchangers in the soil and stimulates release of Al. Finally, Lange et al. (1989) proved in a very elegant experiment that the interaction of N with growth causes Mg deficiency in spruce. Buds were removed or not removed on opposite lateral twigs along the same branch. Damage was observed only on twigs where buds had not been removed and where growth had occurred (Chap. 11, Box 11.4).
Obviously, the growth of trees is significantly regulated by N supply. Thus, canopy N uptake from airborne pollutants (in rain, fog and dew) becomes particularly important because this additional N supply is not balanced by cation uptake but leads to increased growth and the observed yellowing. The model of a nitrogen-cation interaction with limited cation supply due to soil acidification observed for forest decline could also be related to other observed cation deficiencies, particularly K deficiency on bogs, Mn and Fe deficiency on limestone, and the rarer Ca deficiency.
Air pollutants (gases in Fig. 16.26) were thus involved in each of these pathways of decline. The combination of soil acidification, N-triggered growth and ozone together with interactions of insect pests and microbial pathogens caused forest damage across Europe (Last and Watling 1991). Thus, ecological research provided clear evidence on the potential pathways of decline, upon which policy decisions could be taken. Based on scientific evidence, state regulations were put into place controlling emissions from large electric power plants. Thus, sulphur deposition into ecosystems decreased and, in turn, the rate of soil acidification. Additional measures in forest management were taken (liming, substantial clearing), and damaged areas were reforested. Heavy clearing reduced the density of trees in declining forests far below the recommended values of yield tables, for example, in the Fichtelgebirge (Germany) (Fig. 16.27).
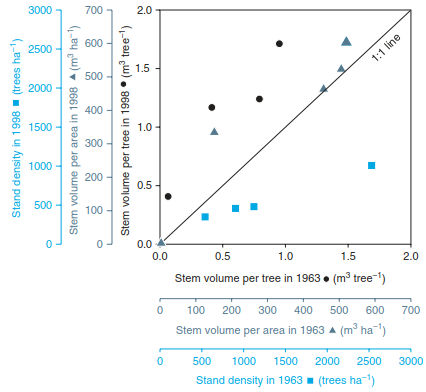
Fig. 16.27. Effects of management on forests. Logging in the 1980s left free-standing single trees. With smaller S, but still high N deposition rates, the remaining individual trees grew faster. Despite fewer trunks and a smaller leaf area, the volume of growth per stand remained the same. (Data from Mund et al. 2002)
Thus, the cation supply per tree increased and, together with the (then) still high N deposition and the higher light availability, growth of individual trees improved. Despite the reduced density of stands, wood growth per area was eventually maintained. Today, damage to spruce and pine stands has been stabilised, but damage to deciduous trees continues to increase.
Date added: 2026-04-26; views: 2;
