Decomposition and Stabilisation of Organic Matter in Terrestrial Ecosystems
If not harvested or eaten by animals, plant tissues naturally senesce and die, become (woody) debris or above- and below-ground litter, and are subsequently decomposed. Decomposition products can be stabilised, contributing to soil carbon pools. Thus, both decomposition and stabilisation of organic matter are tightly linked. Two main processes happening at the same time contribute to decomposition:
- Decay, which describes the biotic breakdown of dead plant organic matter (also called nec- romass) into smaller pieces (degradation) by soil fauna and microorganisms and the related mass loss (Sect. 20.4 in Chap. 20).
- Mineralisation, which describes the release of nutrients in inorganic forms from organic matter, including the release of CH4 or CO2 (Sect. 16.2.1).
Foliage decomposition is strongly linked to litter quality (in particularly to relative concentrations of water-soluble C compounds, cellulose, lignin and N) (Cotrufo et al. 2013) and to environmental conditions. The higher the quality, the faster the breakdown, under adequate soil moisture and temperature regimes for microbial activities (Sect. 20.4, Chap. 20). Simple decay models describe the patterns of exponential mass loss per year (Eq. 16.16). For parameterisation of organic matter decay and SOM formation in ecosystem and global models, Sects. 15.3 and 22.3 in Chaps. 15 and 22, respectively:

with X/X0 describing the percentage mass remaining and к being the decomposition constant (year-1).
Decomposition constants (also called к-values; under steady-state with 1/к = MRT, mean residence time or turnover time) for mass loss range from about 0.25 to 0.47 in Mediterranean and temperate regions vs. 2.3 in tropical areas. Thus, it takes on average 2.1-4 years vs. about 5 months to decompose foliage litter in Mediterranean and tropical climates, respectively. This first step of foliage decomposition occurs in several phases, with the dynamics for C (Fig. 16.14a) being different to those for N (Fig. 16.14b).
Fig. 16.14. Degradation of leaf litter from beech over time. a Initially, the easily available carbohydrates are consumed. Then lignin decomposition starts (after Berg and Matzner 1997). b Parallel with the decomposition of dry matter, decomposition of N-containing substances occurs. (Data from F. Cotrufo)
For a newly fallen leaf (leaf litter), its C content decreases because of the use of easily decomposable C compounds (sugar, hemicellulose, cellulose) by soil fauna and soil microorganisms. At the same time, the N content rises initially, as bacteria and fungi settle on the dead leaf (for beech, this takes about 1 year). Finally, N-containing substances are decomposed, and the N content of the leaf decreases as organisms retreat from the leaf (for beech, after about 3 years). After 3 years, the leaf has lost about 80% of its mass and 60% of its N content (Cotrufo et al. 2000). Further decomposition in later phases is much slower. Lignin is then decomposed by fungi, but for this process an additional C source is required (Gleixner et al. 2001).
Decomposition of wood takes much longer than leaf or fine root decomposition, often between 50 and >100 years. Reported k-values for coarse woody debris (>10 cm in diameter) range from 0.0025 (400 years) to 0.089 (11 years). However, these very long residence times are rather exceptions. In general, mean residence times are shorter in the tropics (8-21 years) than in temperate latitudes (2129 years) or in high latitudes (25-28 years) (Bloom et al. 2016). Mass loss rates vary strongly with wood density and wood chemistry, but also with decomposer activities linked to environmental conditions. Due to higher lignin:N ratios, the wood of gymnosperms decomposes slower than that of angiosperms, when trees grew at the same site (Weedon et al. 2009).
Lignins are high- molecular-mass, three-dimensionally structured compounds of phenylpropane units that enclose cellulose fibrils. Conifers form lignin from coniferyl alcohol and deciduous trees use both coniferyl and sinapyl alcohols. Grasses also use coumaryl alcohol. Degradation occurs by lac- case-forming fungi (white rots), which break bonds in side chains and aromatic rings by forming oxygen radicals (Zech and Kogel-Knabner 1994). Mass loss of wood is also related to the fire history of the site (Fig. 16.15). In boreal coniferous forests, trunks killed by fire are degraded faster than in unburnt areas. Mass loss is finished after about 120 years, with part of the dead wood being burned because of periodic ground fires.
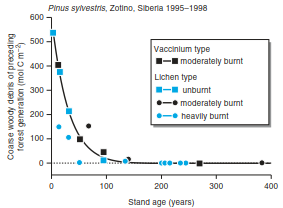
Fig. 16.15. Decomposition of dead wood after a fire in a Siberian pine forest. The mean residence time is only about 40 years, despite the cold climate. This is because of repeated fires at ground level. As these fires only burn the outside of the wood, single dead trunks can be traced back 400 years. (Wirth et al. 2002)
The stabilisation of organic matter, that is, the formation and protection of SOM, has received a lot of attention over the last decade. Stabilisation is related to chemical changes of decomposition products mediated by soil fauna (e.g. earthworms that change the chemistry of the organic residue) and microorganisms, but also to physical protection, for example, by the soil matrix, by association with minerals or occlusion in soil aggregates. Both factors can reduce the chance that organic matter will be decomposed and thus lead to the persistence of organic matter in the soil over long time periods (Schmidt et al. 2011; Cotrufo et al. 2013), contributing to long-term carbon (and nutrient) storage in soils. For example, newly formed microbial polysaccharides (bacterial slime) bind to clay and silt fractions in the soil and are thereby stabilised against further decomposition.
Thus, the clay content determines the ability of soils to store C (Bird et al. 2001). In stark contrast to the initial steps of decomposition, during stabilisation, the molecular structure of the organic matter or the recalcitrance of organic matter as a substrate for decomposition is only marginally important. Even “easily decomposable” sugars can persist in the soil for decades when protected (or immobilised in constantly active microbial biomass), and even “recalcitrant” lignin and plant waxes can be decomposed at rates higher than the bulk soil when conditions are right. Similarly, the relevance of humic substances as very stable SOM fractions, formed de novo during decomposition, needs to be revisited. Inferred from the classical extraction methods in soil chemistry, the amount and relevance of humic substances have been largely overestimated.
Their new formation is no longer considered quantitatively relevant for humus formation in soils. Nevertheless, most soil carbon models still use the structure and inferred decomposability of organic matter to drive their decomposition models (Chaps. 15 and 22). Instead, fire-derived organic matter has been identified as being highly relevant and can make up to 40% of total SOM in many forest and grassland soils. Deep soil layers are still a black box, although globally they store more than 50% of total soil carbon pools.
Date added: 2026-04-26; views: 2;
