Nitrogen Fluxes in Terrestrial Ecosystems
The transformations of nitrogen in terrestrial ecosystems probably correspond most closely to what is generally called a biogeochemical cycle (Fig. 16.22). Elemental nitrogen N does not occur in nature, and most nitrogen is found in the atmosphere as gaseous N2. This N2 cannot be directly used by higher plants unless they are in symbiosis with microorganisms, for example, Fabaceae and rhizobia. In ecosystems, nitrogen occurs in inorganic oxidised and reduced forms (e.g. nitrate NO3- and ammonium NH4+) or together with C in organic compounds, particularly as amino groups (C-NH2) or amide groups (C-N-C). Thus, the C and N cycles are tightly coupled. In what follows, “N” refers not to N2 but to mole equivalents of nitrogen in different oxidised or reduced forms.
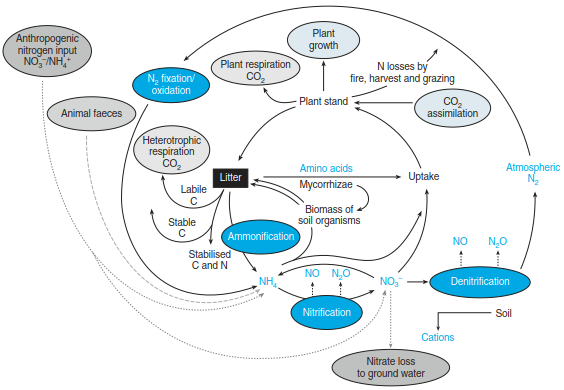
Fig. 16.22. Nitrogen cycle in terrestrial ecosystems with a focus on plant–microbe interactions (Modified from Schulze (2000)). Not all microbial processes are depicted
Overall, the nitrogen cycle in terrestrial ecosystems is characterised by interactions of organisms at different trophic levels (microorganisms, plants, animals, mainly soil fauna and herbivores), multiple origins of the same N species (e.g. NO and N2O) and plant uptake of different N species (NH4+, NO3-, small amino acids). The entry point of nitrogen into ecosystems happens via nitrogen fixation (Fig. 16.23a).
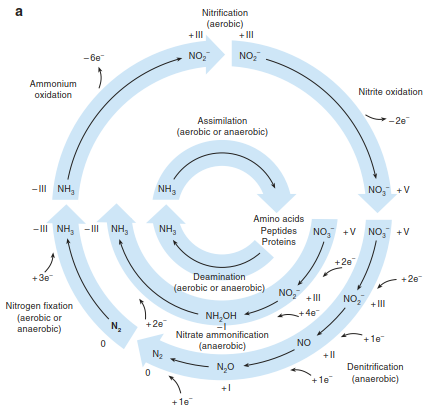


Fig. 16.23. Changes in oxidation states of N during N transformations and impacts of N2 fixation. a Functional groups of microorganisms in biogeochemical nitrogen cycle. Blue arrows indicate reactions that occur within a single organism. Small black arrows are intermediate products. The figure also shows the oxidation state and the uptake or loss of electrons (after Meyer 1994).
b Soil acidification as a result of N2 fixation: During N2 fixation, some plant roots release protons (derived from the dissociation of amino acids in the roots to keep ionic balance), which leads to acidification in many soil types. The low pH in the soil close to the pea root is in contrast to that close to the maize root, which raises the pH by absorbing nitrate. (Photo courtesy E. George).
c, d Macrozamia communis as an example of the symbiosis between a plant and atmospheric N2-fixing bacteria. The cycads form on their hypocotyls so-called coralloid (or coral-like) roots from the cortex that contain cyanobacteria recognisable by the blue-green colouring of roots. Several species of Macrozamia grow in the nutrient-deficient forests of Australia. (Photo: E.-D. Schulze)
The change of gaseous N2 from the atmosphere into organic compounds is achieved by N2-fixing bacteria, which occur as free-living cyanobacteria, as bacteria in nodules of Fabaceae or as symbionts with other higher plants (e.g. alders, cycads plants) (Fig. 16.23b-d) and with fungi (e.g. lichens, biological crusts). Electrons are derived from the degradation of organic substances and used to reduce N2 to NH3. N2 fixation is a process that needs a lot of energy, that is, 16 equivalents of ATP are hydrolysed during this reaction:

N2 fixation is low when N availability in the soil is high (e.g. in agriculture), because plants then use inorganic N forms instead of the energy-intensive N2, or when P availability to the plant is low, because of the high need for ATP (Eq. 16.19). One can see already from this first reaction in the N cycle that the oxidation number of N changes depending on the N compound in question (Fig. 16.23a). In addition to N2 fixation by bacteria, N2 can also be oxidised by lightning or fire.
In the reduction state of NH3, nitrogen can be transformed in many metabolic processes without further changing its state. Only during the transformation of organic substances in the soil are electrons removed from nitrogen via binding to oxygen. A veritable “zoo” of very different soil bacteria is involved to handle the electrons. Distinction is made between the following dominant reactions. Transformation of NH4+ into oxidised nitrogen occurs via ammonification (to nitrite NO2-) (Eq. 16.20) and via nitrification (to nitrate NO3-) (Eq. 16.21):
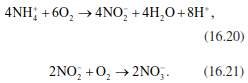
Autotrophic soil bacteria of the genera Nitrosomas (ammonification) and Nitrobacter (nitrification) are responsible for these reaction and use the energy released as energy sources. The real reactions are (even more) complicated (than shown), as for example during the formation of nitrite the oxygen used as electron acceptor comes not from O2 but from water. During the conversion of ammonium to nitrate, losses of intermediate products, particularly the gases NO2, NO and N2O, are also possible (Fig. 16.22). These losses affect the radiation balance of the Earth (Chap. 9).
In addition, microorganisms are able to use the oxidised NO3- as an electron acceptor, thereby gaining the oxygen required for oxidation of other substrates (anaerobic nitrate ammonification, also called nitrate respiration). NO3- thus returns to the reduction state of NO2- (Eq. 16.22) or directly into NH4+ (Eq. 16.23):
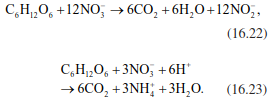
To close the N cycle of an ecosystem, nitrate needs to be transformed into molecular N2 again. This takes place under anaerobic conditions during denitrification. Nitrate serves as electron acceptor. In addition, harvests, fire and leaching cause further N losses (Sect. 16.3). Denitrification is not just one reaction but multiple reactions that can take place in different organisms and that do not necessarily achieve the final product N2. Depending on soil pH, redox conditions and other soil chemical conditions, products of intermediate steps, particularly NO2, NO and N2O, can be released, similar to the release during ammonifi- cation. The total balance of denitrification is
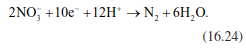
Plants are able to take up different N species, NH4+ as well as NO3- and small amino acids (Chap. 11). For more details on the consequences of N uptake for biodiversity, Sect. 20.4 in Chap. 20. Uptake can be below-ground via roots, but also above-ground. Here, atmospheric N deposition comes into play. Anthropogenic sources of reactive nitrogen include NO*, originating from burning and combustion processes, and NH3, mainly from livestock, fertilisation, sewage systems or industrial production of ammonium. The N deposition in ecosystems from such sources often exceeds 5 kg N ha-1 year-1 (Sect. 21.2, Chap. 21). In some areas with intensive industry or agriculture, N deposition can reach 20 to >50 kg N ha-1 year-1 (Bobbink et al. 2010).
N deposition has been shown to be one of the main drivers for changing species composition in many ecosystems, mainly due to the outcomes of resource competition. In addition, N inputs into ecosystems trigger soil acidification and can (Sect. 16.4). Using stable nitrogen isotope labelling, the fate of N deposition can be followed. It turns out that plant biomass distribution within an ecosystem is not a good indicator of short-term N retention (Buchmann et al. 1996). Understorey vegetation was a larger N sink (9-15%) than 15-year-old Picea abies trees (3-7%), although tree biomass was a factor 4 larger than understorey biomass. The main N sink was the soil (79-87%), particularly the top soil (46-63%). A recent meta-study revealed that soil carbon stocks were a good predictor of 15N tracer retention in 48 ecosystems. Thus, soils represent a large sink for atmospheric N deposition, which can prevent N leaching as long as N deposition stays below approx. 46 kg N ha-1 year-1.
Plant annual N requirements can be met by N mineralisation, N2 fixation and N deposition or fertilisation. But also internal N fluxes, for example, remobilisation of amino acids prior to leaf shedding, can contribute to meeting these N requirements, although the extent varies widely. For example, N concentrations in litter vary between 5 and 10 mg g-1 dry matter in needles and leaves, up to 50 mg g-1 dry matter in crops and forage plants, and between 1 and 5 mg g-1 dry matter in wood. In a temperate beech forest, about 3 t dry matter ha-1 of litter reaches the soil each year, providing an annual input of plant-derived N of about 15-30 kg N ha-1 year-1 for litter decomposition and mineralisation and, thus, in turn for plant uptake. On the other hand, the N requirements of a temperate spruce forest in Germany could not be met by soil N availability alone: about 12% of annual N requirements were met by atmospheric N deposition via above-ground N uptake in the canopy (Horn et al. 1989), compared to 10% in Eastern US conifer sites (Sievering et al. 2000). Nave and Curtis (2011) estimated that about half of the N deposition is intercepted and taken up in forest canopies.
The N supply via N mineralisation (i.e. ammonification and nitrification) is highly variable with respect to both the chemical species and the amount. At very low supply (N deficiency) and in acidic soils, fungi are the dominant microorganisms in the soil (Smith and Read 2008). They can make organic nitrogen available via proteases, which are particularly active at low pH, that is, fungi acidify the substrate by releasing protons and thus directly take up organic nitrogen from litter. Fungi generally have a higher N requirement than plants since in several species the cell walls are formed by N-rich glucosamine (chitin).
Fungi also require carbohydrates that they obtain as mycorrhizae either directly from plants or as saprophytes living on organic substances from the decomposition of litter and debris. Mycorrhizal fungi supply amino acids derived from protein degradation to the plant, in exchange for carbohydrates. In boreal coniferous forests, this “short-circuited” nitrogen cycle, bypassing mineralisation by microbes, is so effective that no free nitrate or ammonium may be found in the soil solution (Wallenda et al. 2000). Despite the dominance of fungi in the degradation of organic substances and in the uptake of N in boreal forests, there are also ammonium-forming and nitrifying bacteria, as shown by the presence of spores that become active after long incubation times (Persson et al. 2000).
Ammonium and nitrate only occur in soil solutions in boreal climates when the supply of calcium is high, so the soil pH increases (Nordin et al. 2001). Bacteria are more effective than fungi at mineralizing organic matter at higher pH values. Under these conditions, nitrate and ammonium can be detected in the soil solution as the main N products of decomposition (Sect. 16.2). In soil, oxidation and reduction of nitrogen compounds can take place concurrently in all horizons, since the inside of soil aggregates provide oxygen-free (anaerobic) zones.
An excess of ammonium or nitrate, also termed nitrogen saturation, occurs when external supply (by N deposition, fertilisation) or formation in the ecosystem (by mineralisation) exceeds consumption (e.g. by seasonal variation of growth). Nitrate leaching of 5 kg N ha-1 year-1 is used as an indicator for N saturation thresholds. This has very significant consequences for the ecosystem (Sect. 16.4). Ammonium excess causes the release of cations, particularly K+ and Al3+ from clay minerals (Chap. 11). In contrast, the highly mobile nitrate ion is not bound to the soil exchanger. Thus, nitrate excess can lead to nitrate leaching to lower soil horizons, but also to groundwater and, further, drainage systems.
This loss of anions is coupled to an equimolar loss of cations. Deposition of N from air pollutants often accelerates N transformations and leads to increased nitrate loss, even without interaction with organisms in the ecosystem (Durka et al. 1994; de Vries et al. 2003). As a result of high N deposition rates and negative environmental effects, the critical load concept was developed in the 1980s. It calculates a threshold of N deposition for ecosystems below which one does not expect any negative effects. The set critical load for most temperate forests is between 10 and 20 kg N ha-1 year-1. Over time, many studies have now identified the real critical loads. They range from 5 to 10 kg N ha-1 year-1 for boreal forests, tundra, bogs and alpine ecosystems to 20-30 kg N ha-1 year-1 for low- and medium-elevation hay meadows.
The highest critical loads, 30-40 kg N ha-1 year-1, have been reported for salt marshes. Still, more than 25% of all European forests suffer under N deposition rates higher than their corresponding critical loads. However, long-term fertilisation trials in Swedish forests contradict the notion that high N deposition directly translates to N leaching. Despite N fertilisation rates between 20 and 100 kg N ha-1 year-1 (as ammonium nitrate, adding up to about 2000 kg N ha-1 over 30 years), no N leaching occurred; instead N additions were used for increased forest growth (Binkley and Hogberg 2016). Magnani et al. (2007) found a very strong positive relationship between net ecosystem production (Sect. 16.2) and wet N deposition (<15 kg N ha-1 year-1) and questioned the risk of widespread N saturation under natural conditions. However, they were heavily criticised for their assumptions (de Vries et al. 2008).
Date added: 2026-04-26; views: 2;
