Functional Diversity and Plant Traits: A Trait-Based Approach to Ecology
“In the quest to understand species coexistence and community assembly, and to address the ecological consequences of anthropogenic changes, ecologists have moved from counting species to accounting for species” (Cadotte et al. 2013, p. 1234, italics added). This insight derives from the fact that species largely differ ecologically and play different roles or functions in ecosystems. That is, besides species richness, evenness and composition, the ecological dissimilarity or functional differences among species cover an important aspect of biodiversity. To describe the dissimilarity among biological entities with respect to their functional roles in ecosystems, different measures of functional diversity have been developed. Functional diversity relates to “those components of biodiversity that influence how an ecosystem operates or functions” (Tilman 2001). It has increasingly been used to study the consequences of biodiversity change for ecosystem functioning and the delivery of ecosystem services because it includes information on species characteristics—or traits—that are assumed to control ecological processes (Sect. 20.4).
In essence, any measure of functional diversity will summarise the value, range, distribution and relative abundance of traits in a community. In other words, indices of functional diversity capture the amount of variation in a multivariate trait space or hypervolume represented by the species (or other biological entities) within a community. This trait hypervolume thus characterises the phenotypic space occupied by a set of species. Therefore, it is important to get a clear understanding of the meaning of functional traits. Traits are characteristics of plants at the scale of cells, tissues and up to the whole organism that reflect their evolutionary history and that shape their performance. Hence, traits are rather broadly defined and encompass heritable quantitative morphological, anatomical, biochemical, physiological or phenological properties of organisms (Garnier et al. 2015). These properties must be measurable at the individual level. Traits can be continuous (e.g. specific leaf area, seed size) or categorical (e.g. life form, leaf habit).
They have a direct or indirect impact on the fitness of an individual plant through their effects on growth, reproduction and survival, which constitute the three components of plant performance. Thus, traits offer insights into questions such as (Reich 2014): How and why does a plant “behave” as it does? Where does a plant grow and where does it not grow? How does a plant interact with other plants or other organisms, such as herbivores? How does it influence the abiotic and biotic environment around it? Analysing such questions from the perspective of plant attributes has therefore been called a trait-based approach to plant ecology, based on the work of Humboldt, Schimper, Larcher and many others.
Many traits are often correlated or covary across species or are similar in their functional consequences. These trait syndromes capture fundamental trade-offs along several important axes of plant strategy and function. Three dimensions of plant ecological strategies are considered fundamental for understanding plant functioning: the acquisition and use of resources, the stature of the plant and the capacity for sexual reproduction. For above-ground parts, these dimensions are represented by the leaf economics, canopy size and seed size spectrum (leaf-height-seed scheme) (Westoby 1998). For example, plants may have “cheaply constructed” leaves, with low leaf mass per area (LMA) (kg m-2) resp. high specific leaf area (SLA) (m2 kg-1), that have a short lifespan but high N and P concentrations and gas exchange rates. Among trees, European aspen (Populus tremula) would be an example. At the other extreme of this range, plants invest more biomass per leaf area (high LMA, low SLA), resulting in leaves with long lifespans, often associated with high concentrations of secondary metabolic compounds but low N and P concentrations and gas exchange rates. Norway spruce (Picea abies) would be an example for this strategy.
Plant stature is related to the competitive ability of species: being larger than neighbours confers a competitive advantage in light capture and is therefore an important characteristic of a carbon acquisition strategy. European beech (Fagus syl- vatica), for instance, can grow in the shade of neighbouring trees for a long time, but it ultimately outcompetes other species owing to its large maximum height at maturity. The seed-size trade-off involves plants with large individual seed sizes but a low number of seeds or seed output per canopy area; such plants tend to have higher seedling survival under intense competition or low resource availability. An example would be sweet chestnut (Castanea sativa). Such species follow a so-called K strategy, living in densities close to carrying capacity (K) and producing fewer seeds. In contrast, plants with small seeds can produce many more seeds with the same relative investment, enhancing dispersal to sites with low competition or high resource availability. An example of such a species with high growth rates (r) and high seed output (r strategy) would be silver birch (Betula pendula).
In addition to these three major axes of ecological strategy, other trait dimensions with large variation are also important for plant functioning, including xylem hydraulic and mechanical property trade-offs of stems or wood, which is especially important for tree performance (Westoby and Wright 2006). In trees, the xylem is mainly responsible for the transport of water and nutrients, but also for mechanical stiffness. Therefore, a trade-off between conductive efficiency and mechanical strength can be found. In addition, conductance also trades off with resistance to embolism (i.e. the formation of gas bubbles in vessels, blocking the movement of water) because larger vessels or wider pit pores have a higher risk of embolism.
More recently, the idea of a general whole-plant fast-slow or acquisition-conservatism spectrum of plant economics has been developed. It states that rates of resource acquisition and processing are converging for roots, stems or leaves owing to strong selective pressure along resource axes (Reich 2014). In other words, plant species that are fast in acquiring carbon (leaves), water or nutrients (roots) must also have characteristics enabling fast rates or use at other organs. For example, species that are capable of moving water rapidly also have low tissue density, short tissue lifespans and high rates of resource acquisition and flux at organ and individual scales. The converse is generally found for species with a slow or “conservatism” strategy. The fast-slow spectrum of traits also scales up to the ecosystem level, where the dominance of species with a fast strategy is associated with faster rates of ecosystem processes such as productivity or decomposition of organic matter, and vice versa.
Root traits have been investigated less, but they are of course essential for water and nutrient uptake, anchoring, storage and competitive ability. Interestingly, some below-ground traits seem to covary with above-ground traits in some species, such as rooting depth and maximum height or specific root length (SRL) and SLA. However, for other species, coordination between above- and below-ground traits has not been found, which can be explained by the very different nature of the above- vs. below-ground environment and the different functions of roots, stems and leaves: roots must acquire water and nutrients from the soil solution, stems must provide mechanical strength for height growth and transport, and leaves must capture light and allow gas exchange for photosynthesis. In addition, the presence of mycorrhizae in most plants also changes many root traits, which makes trait coordination between above- and below-ground traits even less likely.
Traits strongly determine the ecological role of plants within a community through two different means. First, they can explain individual plant responses to environmental factors, thereby influencing occurrence, community structure and diversity. Such properties have been called response traits; plant tolerance to frost would be an example, which strongly determines survival and, thus, altitudinal patterns of species occurrence. In addition, traits also determine how plants affect other trophic levels or influence ecosystem processes, that is, they are linked to the capture, use and release of resources. These properties are called effect traits; an example would be the concentration of aromatic substances in leaves, which affects herbivory or decomposition. These two “roles” of traits are also illustrated in a conceptual graph that structures the entire Chap. 20 (Fig. 20.1). It also depicts the central role of plant traits in linking the environment to plant diversity and to ecosystem function. It must be noted that response traits and effect traits may have varying degrees of overlap: for instance, SLA is usually high in nutrient-rich habitats (response trait), and it positively affects whole-plant relative growth rate (RGR) and, thus, also community productivity and biogeochemical cycles (effect trait) (Sect. 12.3, Table 20.4). Species’ tolerance to environmental factors, stressors or disturbances and their contributions to ecosystem functions and services will often not only depend on single traits, but on a combination of traits and often show a phylogenetic signal (Diaz et al. 2013).
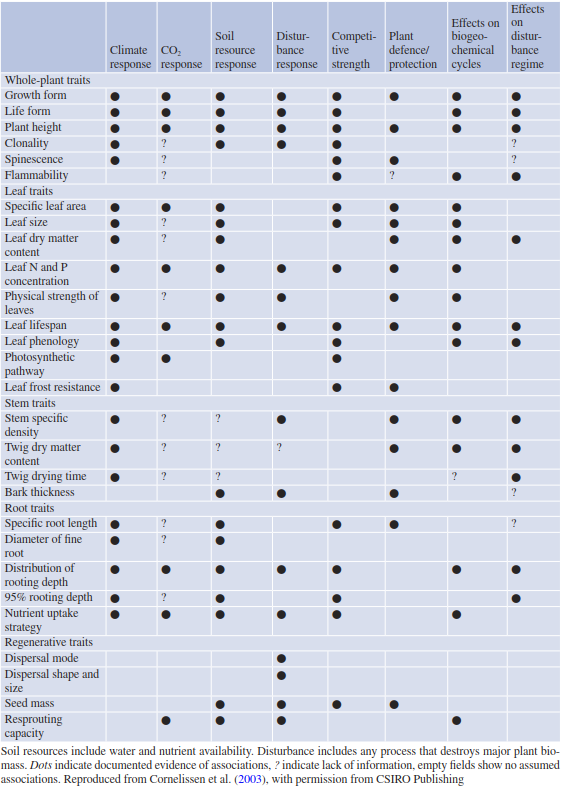
Table 20.4. Association of selected traits with plant responses to environmental changes, plant competitive strength and protection against herbivores and pathogens, and effects on biogeochemical cycles and disturbance regimes
Is it possible to predict certain properties of an ecosystem by the dominance of species with specific traits or by the mean trait value of the cooccurring species (Sect. 20.4.1)? To evaluate this hypothesis, community-weighted means (CWM) of single traits are calculated based on measured trait values, weighted by the abundance or biomass of the species. Community net primary productivity, litter decomposition rates or the accumulation of carbon and nitrogen in the soil, for instance, can be highly predicted by few leaf traits only, for instance (Table 20.5), which are of course correlated with other traits (e.g. root traits) that may have more direct effects on the processes studied. Mean trait values do not represent a measure of functional diversity but are a useful and widely used indicator of the functional structure of a community.
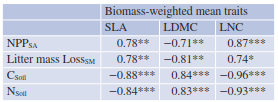
Table 20.5. Predicting ecosystem properties by plant traits. Pearson correlation coefficients between SLA, that is, the ratio of water-saturated leaf area to leaf dry mass (m2 kg−1), leaf dry matter content (LDMC), that is, the ratio of leaf dry mass to water-saturated fresh mass (mg g−1), or leaf nitrogen concentration (LNC) (mg g−1), with specific above-ground net primary productivity (NPPSA), that is, net primary production expressed on a per gram green biomass basis (g kg−1 day−1), specific rate of litter mass loss (Litter Mass LossSM, that is, decomposition rate expressed per unit biomass (g kg−1 day−1), total soil organic carbon concentration (CSoil) (g kg−1) and total soil nitrogen concentration (NSoil) (g kg−1). Biomass-weighted mean traits were calculated as aggregated on a community level. *p < 0.05; **p < 0.01; ***p < 0.001 (Garnier et al. 2004)
Plants are immobile, meaning they cannot physically escape predators and pathogens, search for pollinators or hide from approaching extreme climatic situations. Obviously, plants as sessile organisms must continuously cope with changing environmental conditions—from minute-by-minute, daily, seasonal to decadal fluctuations in temperature and light, changes in nutrient and water availability, or herbivore pressure, pathogen load or loss of mutualistic partners. Plants have therefore evolved a variety of mechanisms that enable them to tolerate and withstand environmental change, and to reachieve internal homeostasis: plants are highly plastic and resilient to cope with a highly dynamic environment.
This phenotypic plasticity, or the capacity of a given genotype to produce different physiological or morphological phenotypes in response to different environmental conditions, is also reflected in plant traits. Traits are usually quantified comparatively across species and are highly context-dependent. This means that traits are not only genetically fixed and heritable properties, but are also plastic. They can change over time (e.g. seasonal or ontogenetic variations) and can depend on environmental conditions, the presence of competitors or herbivores and pathogens. Examples of this phenotypic plasticity of traits are the development of sun and shade leaves within a single crown (Sect. 3.2) or the increase in defensive metabolites after herbivore attack (Sect. 8.3). In fact, plasticity can be considered a trait of its own, given its large variability across species and habitats. Trait plasticity, however, is also limited by multiple ecological factors, for example, extreme levels in a given abiotic factor can reduce plasticity to another factor (Valladares et al. 2007).
The traits present in a plant community also reflect the interplay between evolutionary and assembly processes and the physical environment (Sect. 20.3.2). Thus, understanding how and why plant traits vary among species and sites is a critical step towards understanding ecosystem properties and their functioning. Hence, trait-based ecology has received much attention over the last decade to acquire a better understanding of how traits influence species distribution, interactions and functions (e.g. Garnier et al. 2015 for a recent synopsis with extensive literature). Global data sets covering hundreds of traits and thousands of species are available (Kattge et al. 2011), and handbooks about measurements of traits are useful for standardisation and cross-study comparisons (e.g. Perez- Harguindeguy et al. 2013).
Once traits and their respective associations with ecological responses or effects have been clarified, different measures of functional diversity can be determined. A very critical aspect for its determination is the need to select those traits that are relevant for the process or function of interest, which thus must be defined explicitly. In general, measures of functional diversity fall into two main classes: (1) discontinuous measures, that is, categorising species into plant functional groups or types, and (2) continuous measures, that is, measuring the spread of species in an я-dimensional trait space.
Date added: 2026-04-26; views: 2;
